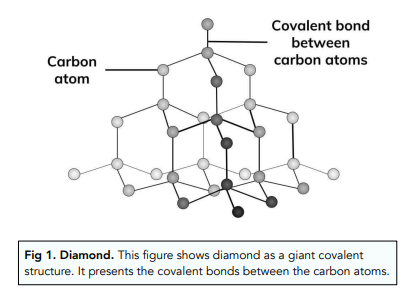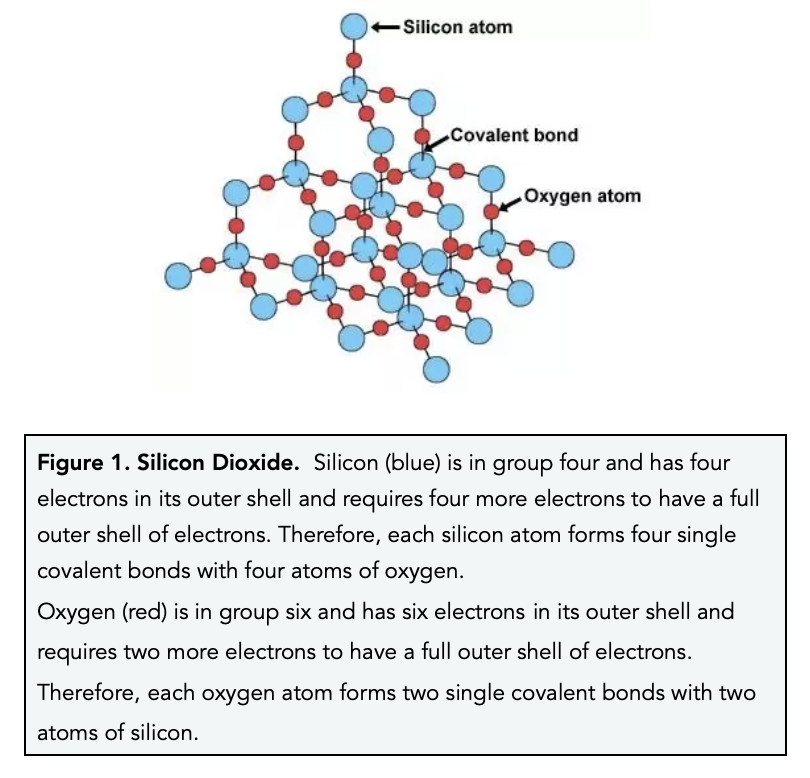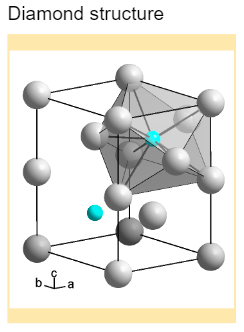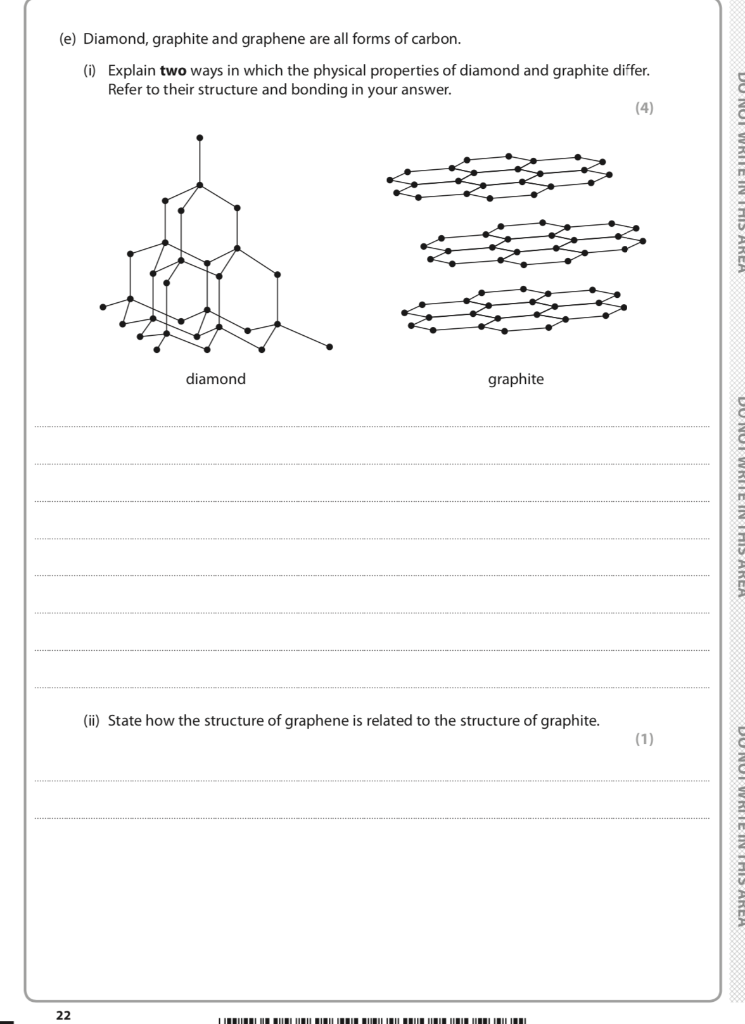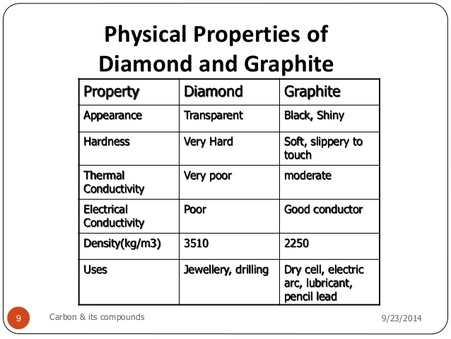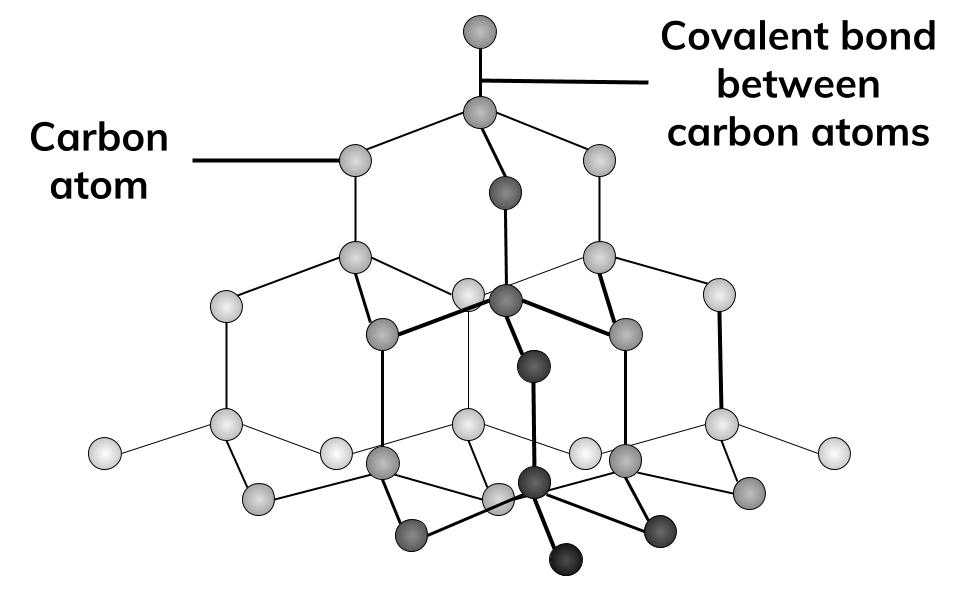
1:50 explain how the structures of diamond, graphite and C60 fullerene influence their physical properties, including electrical conductivity and hardness - TutorMyself Chemistry

Physical Property of Matter Overview & Examples | What is a Physical Property of Matter? - Video & Lesson Transcript | Study.com

Physical & Chemical Properties of Diamonds and their Inclusions: Spectroscopic and Stable Isotope Analyses | Planetary Geoscience Institute

Why diamond and graphite have different physical properties but same chemical properties? What is the property called? - Quora

Physical & Chemical Properties of Diamonds and their Inclusions: Spectroscopic and Stable Isotope Analyses | Planetary Geoscience Institute

SOLVED:Classify each of the following properties as chemical or physical: (a) hardness of a diamond; (b) combustibility of gasoline; (c) corrosive character of an acid; (d) elasticity of a rubber band; (e)

Physical & Chemical Properties of Diamonds and their Inclusions: Spectroscopic and Stable Isotope Analyses | Planetary Geoscience Institute