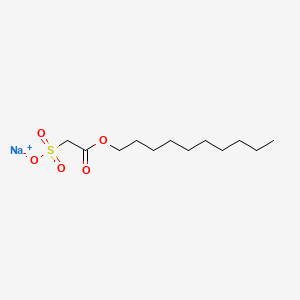
![CAS: 215789-45-0 | trans-2-[4-[(tert-Butoxycarbonyl)amino]cyclohexyl]acetic Acid Methyl Ester | SynThink CAS: 215789-45-0 | trans-2-[4-[(tert-Butoxycarbonyl)amino]cyclohexyl]acetic Acid Methyl Ester | SynThink](https://synthinkchemicals.com/wp-content/uploads/2021/10/SA39702.png)
CAS: 215789-45-0 | trans-2-[4-[(tert-Butoxycarbonyl)amino]cyclohexyl]acetic Acid Methyl Ester | SynThink
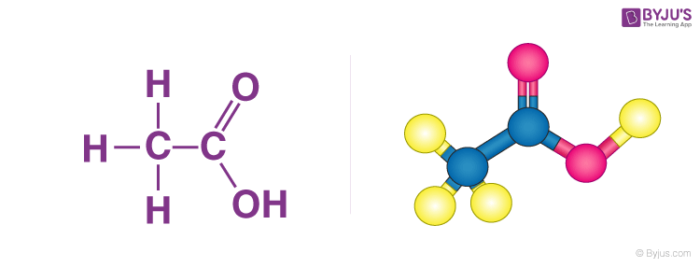
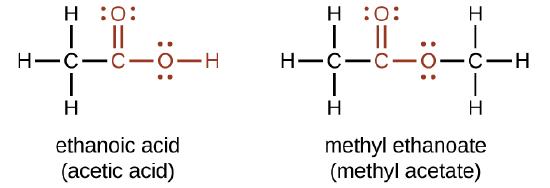
Acetic Acid, Systematically Named Ethanoic Acid, Main Component Stock Illustration - Illustration of ethanoic, skin: 84139031

Synthesis of Enantiomerically Pure 5-Substituted Piperazine-2-Acetic Acid Esters as Intermediates for Library Production | The Journal of Organic Chemistry
CAS#:1233361-76-6 | L-(+)-tartaric acid salt of α-amino-(2-chlorophenyl)acetic acid methyl ester | Chemsrc

Processes | Free Full-Text | Novel Process for Butyl Acetate Production via Membrane Reactor: A Comparative Study with the Conventional and Reactive Distillation Processes
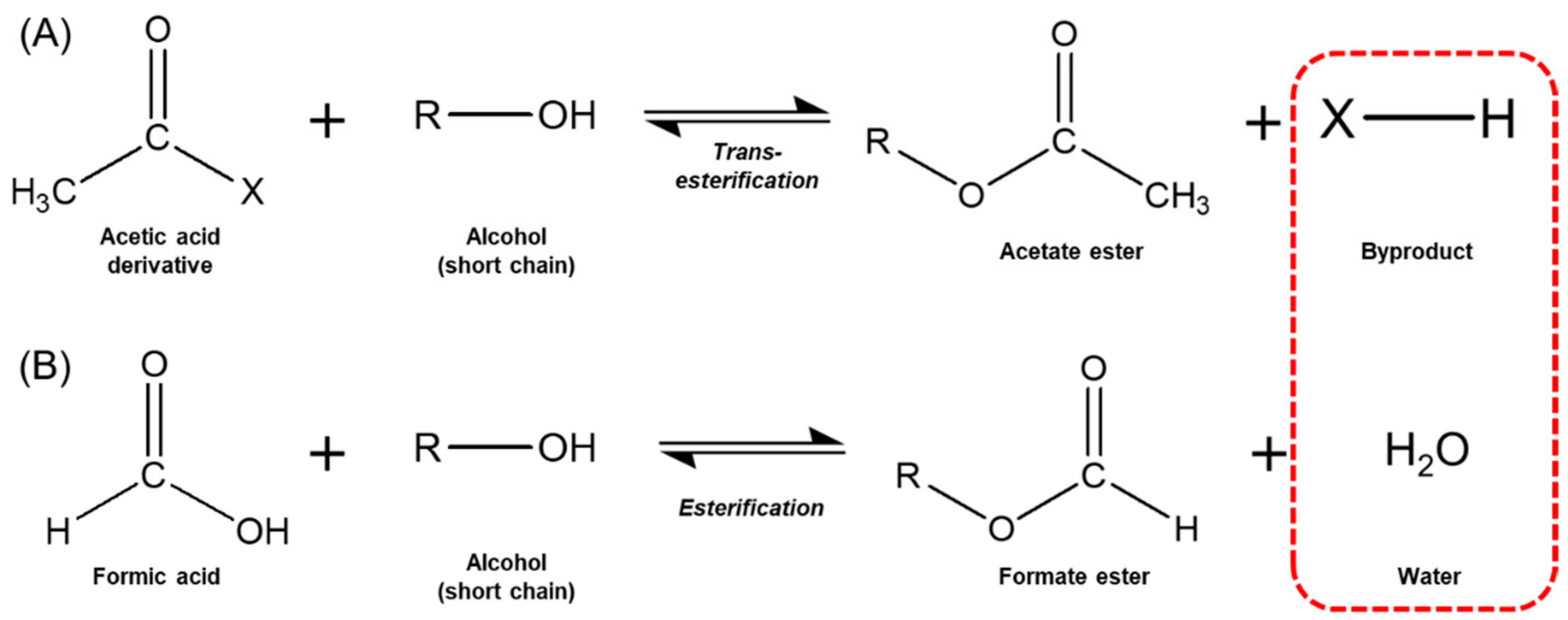
Biomolecules | Free Full-Text | Novel and Efficient Synthesis of Phenethyl Formate via Enzymatic Esterification of Formic Acid

Explain why monoalkylated and dialkylated acetic acids can be prepared by malonic ester synthesis, whereas trialkylated acetic acids (R3CCO2H) can't be prepared. | Homework.Study.com





%E2%82%82.jpg)