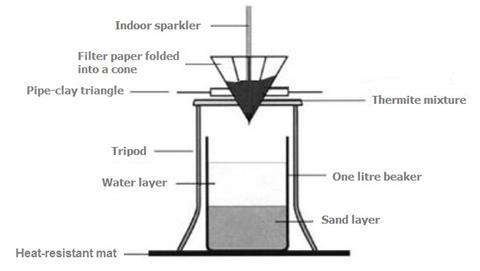
SOLVED: Fe2O3(s) + 2Al(s) –> Al2O3(s) + 2Fe(s) Mass of iron oxide powder = 2.63g Mass of aluminium powder = 0.83g Using the given masses of reactants above, which reactant is the
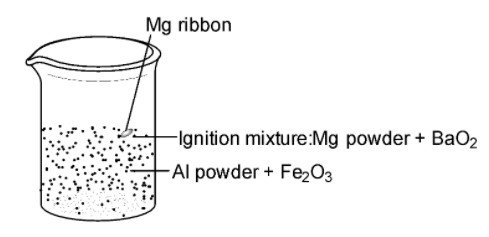
![Thermite By Jack Rjeili. What is Thermite? Mix of aluminum powder and iron oxide [1] Once ignited, produces extreme temperatures Produced from union. - ppt download Thermite By Jack Rjeili. What is Thermite? Mix of aluminum powder and iron oxide [1] Once ignited, produces extreme temperatures Produced from union. - ppt download](https://images.slideplayer.com/33/8235959/slides/slide_4.jpg)
Thermite By Jack Rjeili. What is Thermite? Mix of aluminum powder and iron oxide [1] Once ignited, produces extreme temperatures Produced from union. - ppt download

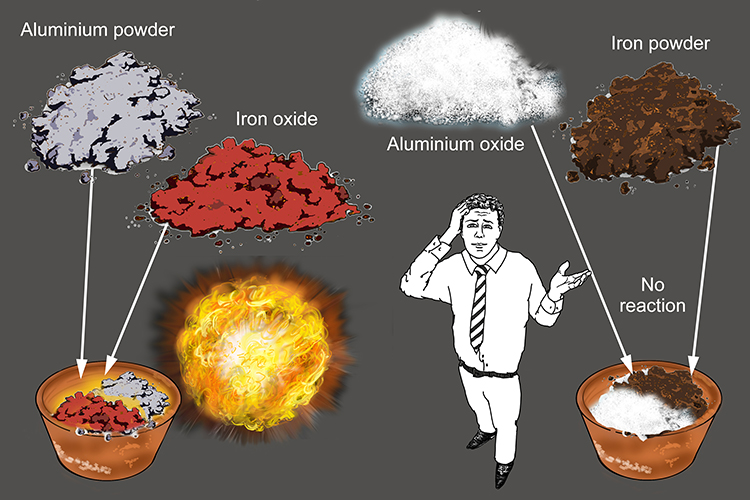
A thermite reaction (iron (III) oxide & aluminium powder) in a UK secondary school (No 2 of 4 image sequence)-see notes for details Stock Photo - Alamy

when a mixture of aluminium powder and iron (III) oxide is ignited, it produces molten iron and - YouTube









:max_bytes(150000):strip_icc()/GettyImages-578813868-5a787515a9d4f90036eff588.jpg)