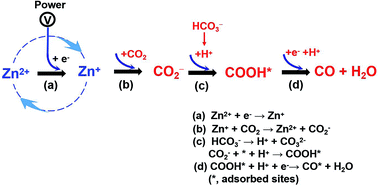
Efficient CO2/CO Separation by Pressure Swing Adsorption Using an Intrinsically Nanoporous Molecular Crystal | ACS Applied Nano Materials

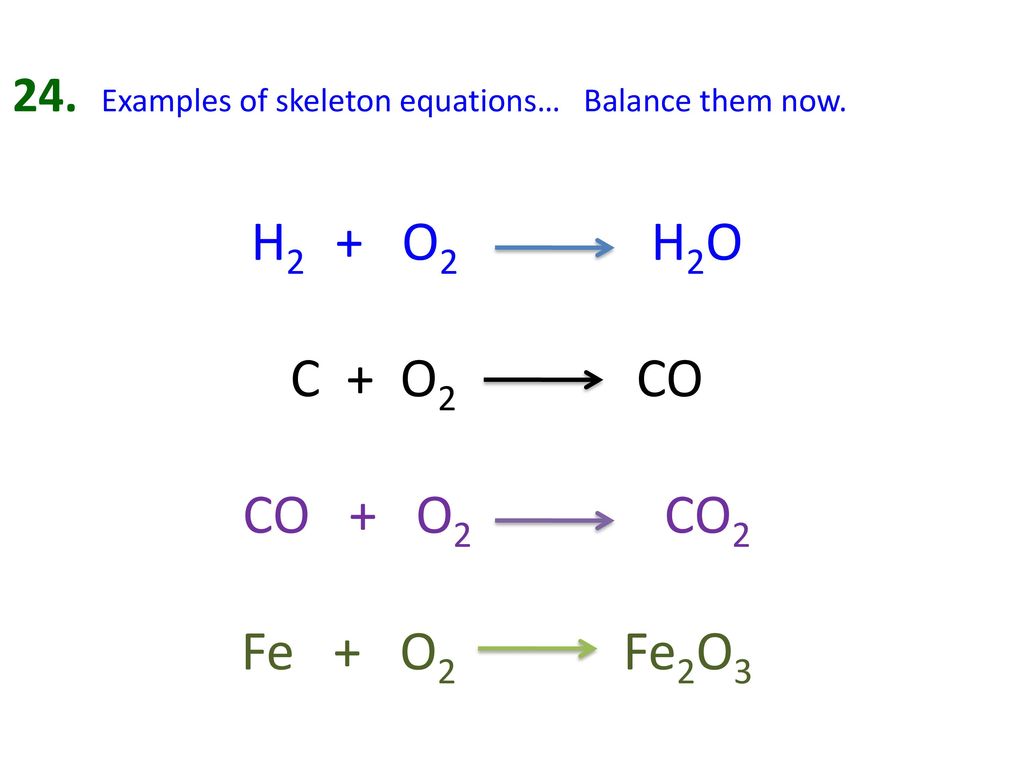
Chemical Equations Section 7.2 & 7.3. Chemical Equations CH 4 (g) + O 2(g) CO 2 (g) + H 2 O (g) Reactantsproducts Means to produce solid (s) Liquid. - ppt download
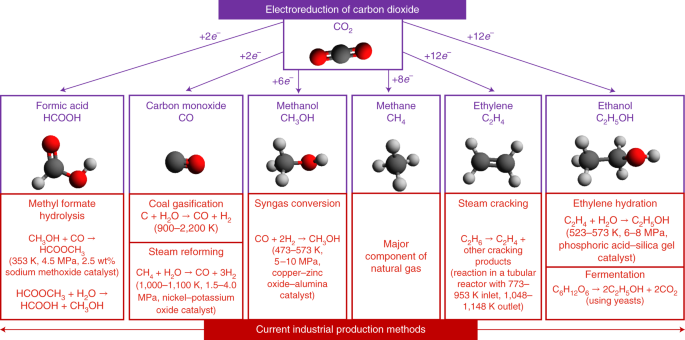
Co-electrolysis of CO2 and glycerol as a pathway to carbon chemicals with improved technoeconomics due to low electricity consumption | Nature Energy
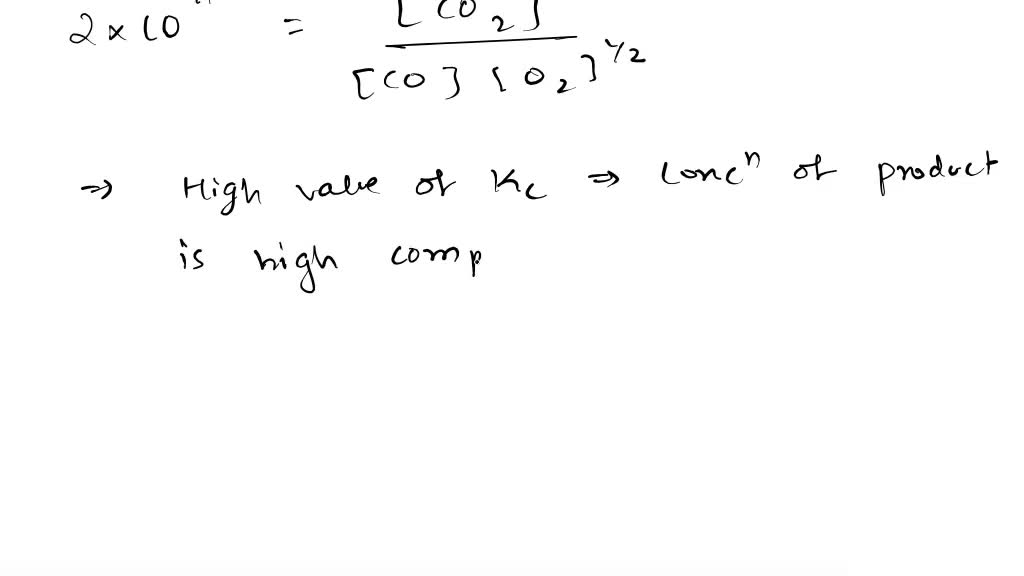
SOLVED: 1. The equilibrium constant for the production of carbon dioxide ( CO2) from carbon monoxide (CO) and oxygen (O2) is Kc = 2 × 1011. A. Write, and balance, the equation for

Efficiency and selectivity of CO2 reduction to CO on gold gas diffusion electrodes in acidic media | Nature Communications
![CO2 = CO + [O]: recent advances in carbonylation of C–H bonds with CO2 - Chemical Communications (RSC Publishing) CO2 = CO + [O]: recent advances in carbonylation of C–H bonds with CO2 - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/D0CC00547A)
CO2 = CO + [O]: recent advances in carbonylation of C–H bonds with CO2 - Chemical Communications (RSC Publishing)
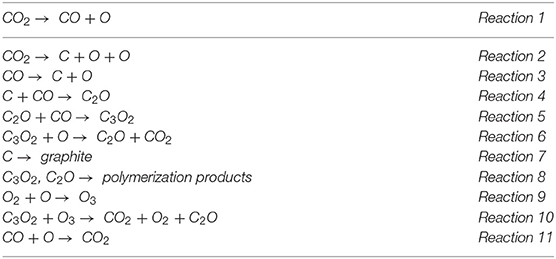
Frontiers | Radiation-Induced Chemistry of Carbon Dioxide: A Pathway to Close the Carbon Loop for a Circular Economy