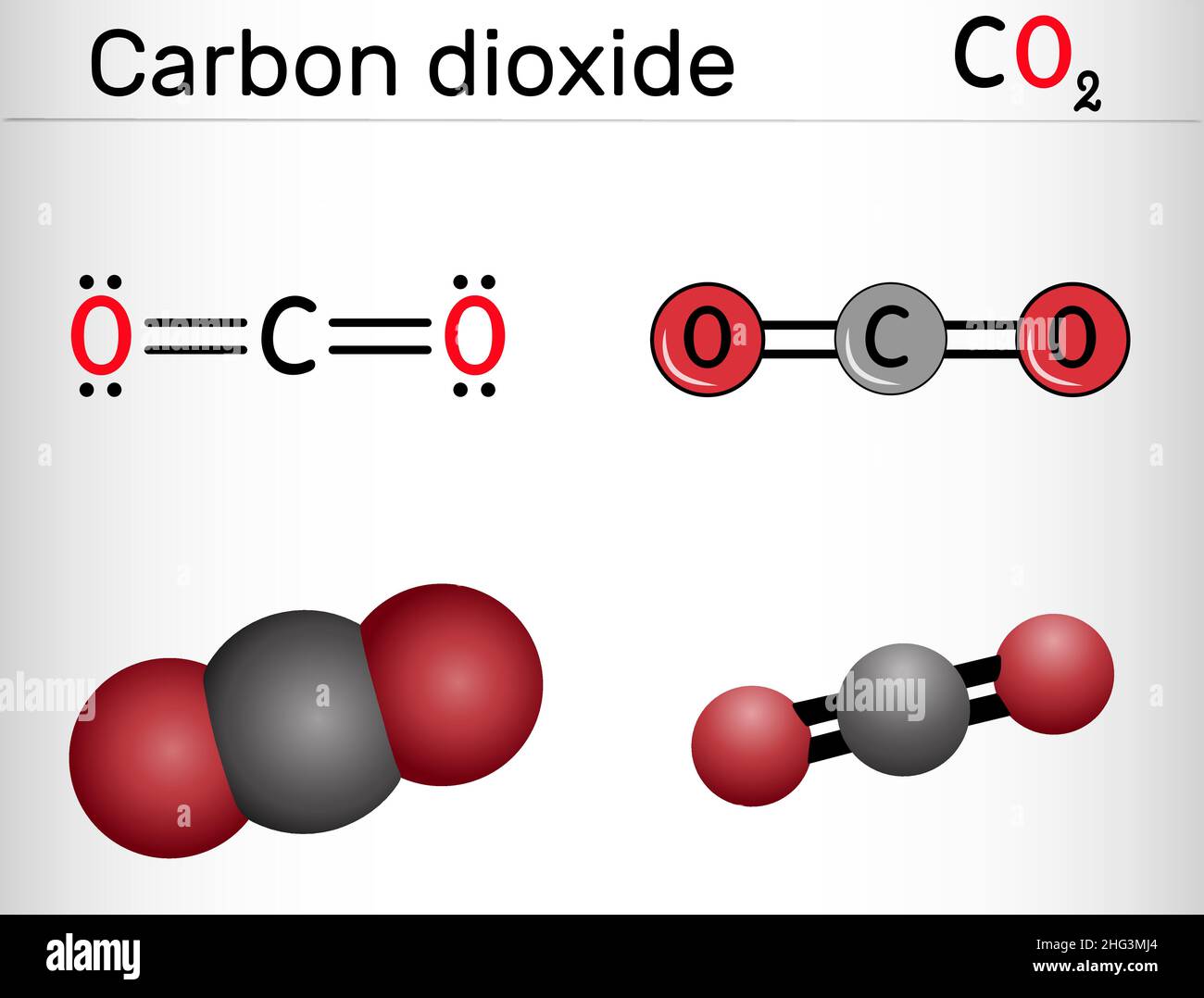
Carbon dioxide, CO2 molecule. It is carbonic anhydride, one-carbon compound in which carbon is attached to each oxygen atom by double bond. Structural Stock Vector Image & Art - Alamy
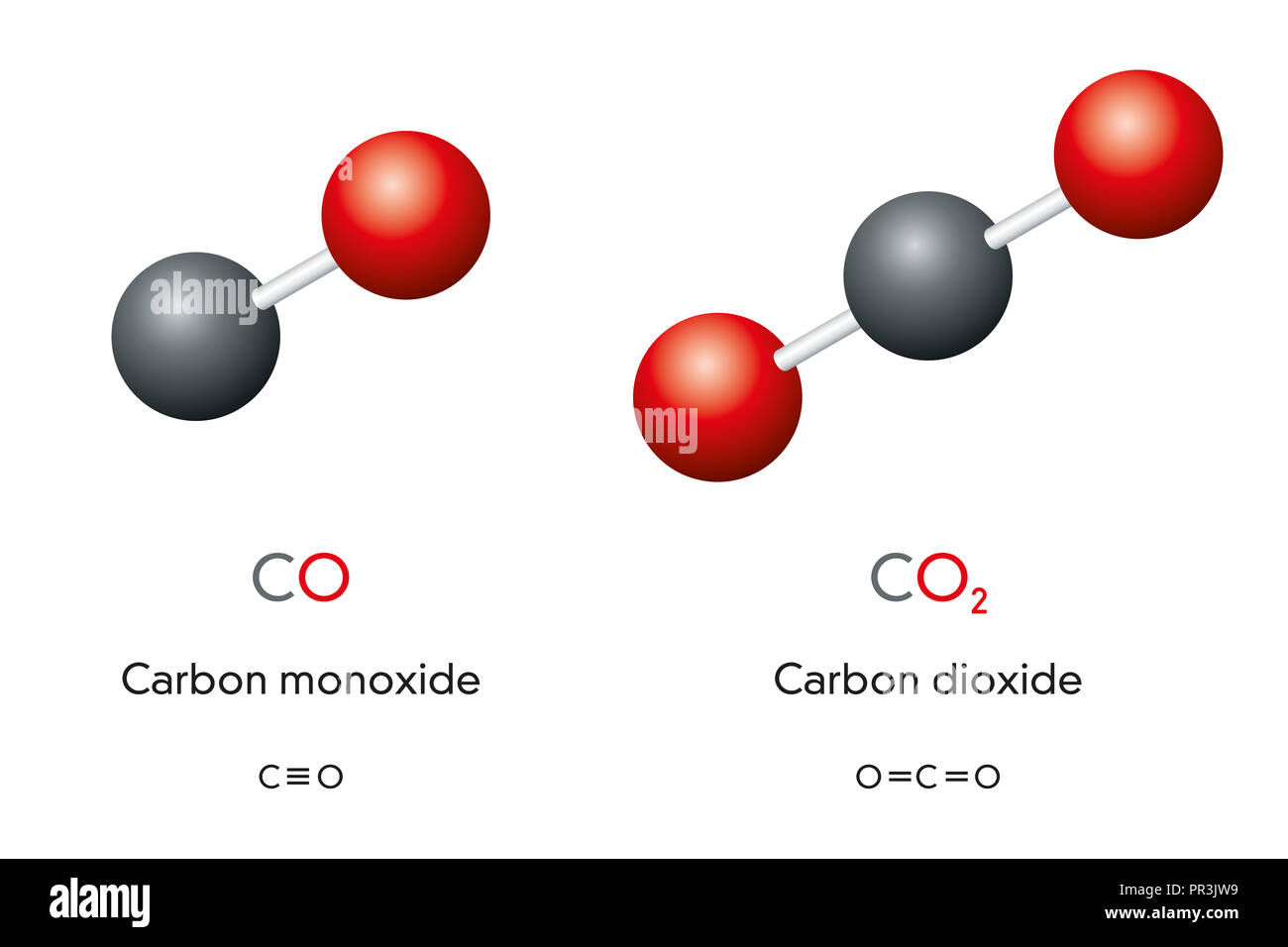
Carbon monoxide CO and carbon dioxide CO2 molecule models and chemical formulas. Gas. Ball-and-stick models, geometric structures and formulas Stock Photo - Alamy

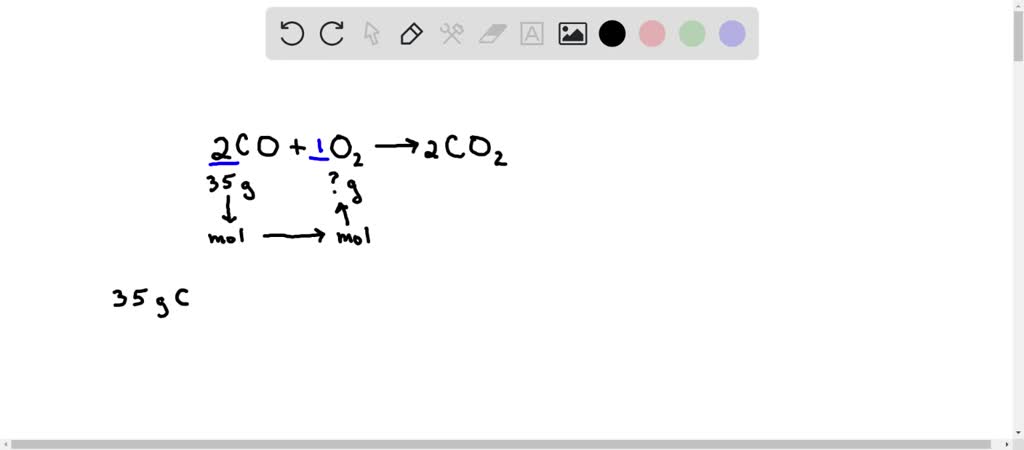
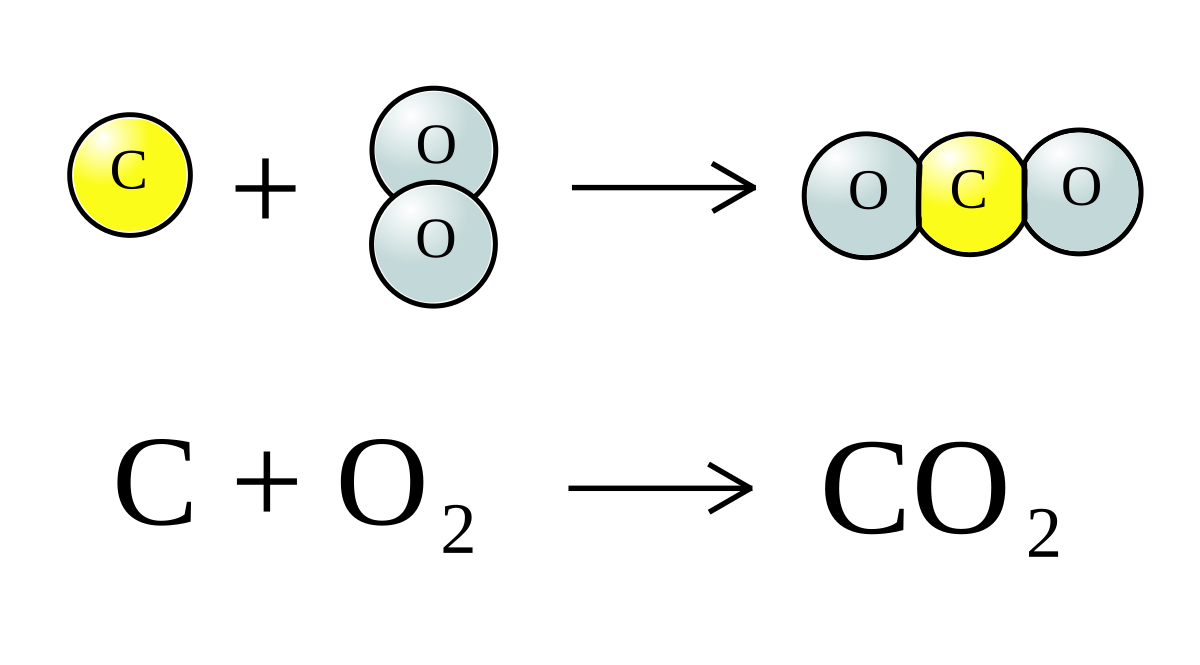
Consider the following reactions, C (s) + O2 (g) → CO2 (g), Δ H = - 94 kcal 2CO (g) + O2 → 2 CO2 (g) , Δ H = - 135.2 kcalThen, the heat of formation of CO (g) is: