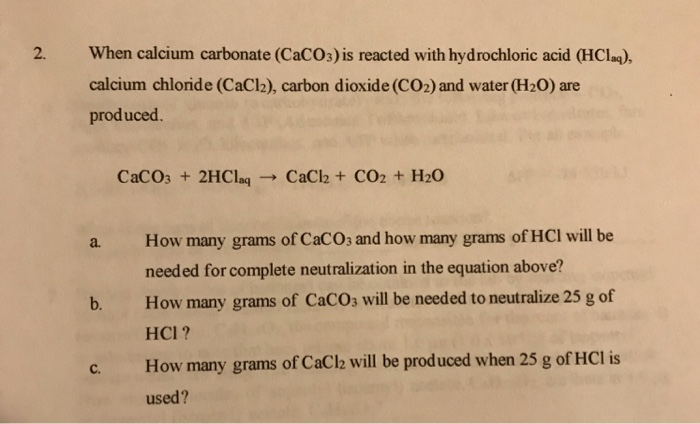Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa

CaCO3 + 2HCl → CaCl2 + H2O + CO2 The mass of calcium chloride formed when 2.5 g of calcium carbonate is dissolved in excess of hydrochloric acid is:

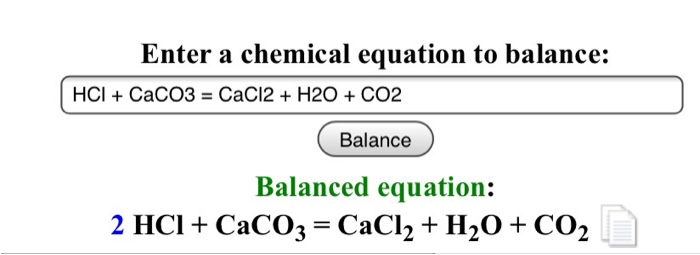
HCl+CaCO3=CaCl2+H2O+CO2 balance the chemical equation @mydocumentary838. hcl+caco3=cacl2+h2o+co2 - YouTube

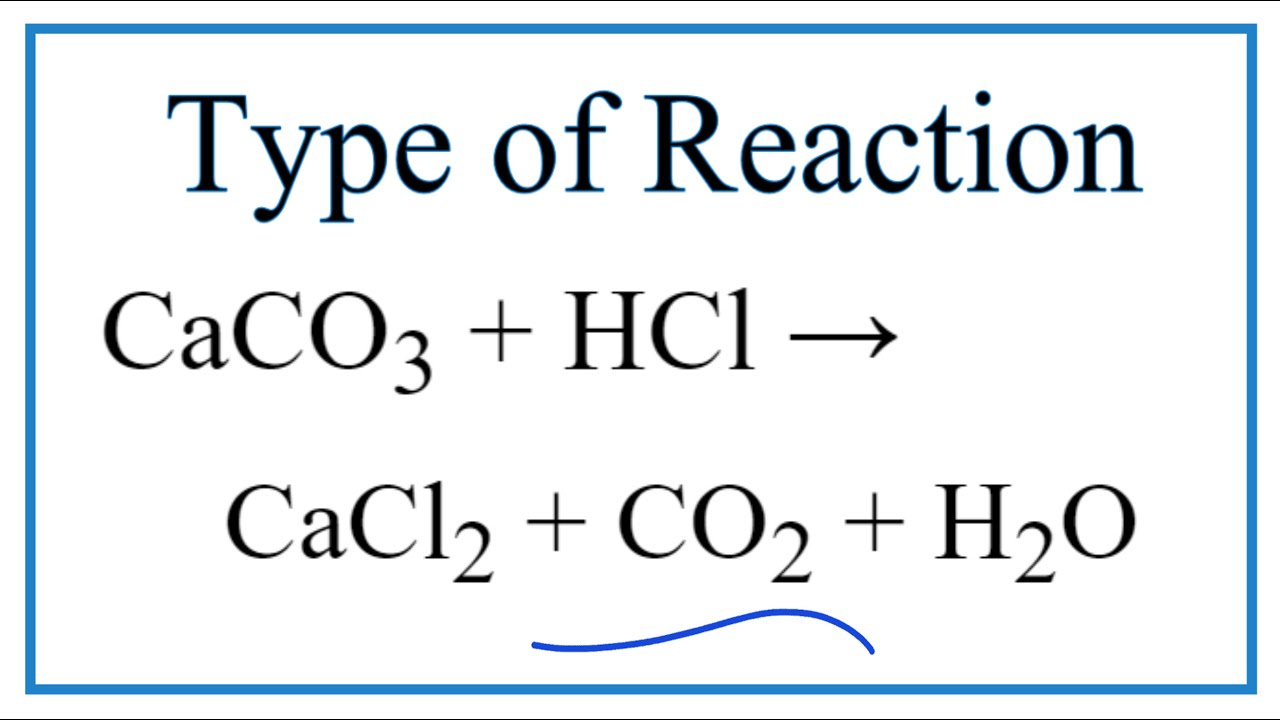
Q2. calcium Carbonate Reacts With Aqueous Hcl To Give Cacl2 And Co2 According To The Reaction Given Below: Caco3 (s) + 2hcl (aq) → Cacl2(aq) + Co2(g) + H2o(l) What Mass Of

Integration of CaO/CaCO3-CaCl2 thermochemical energy storage system with solid oxide iron-air redox battery - ScienceDirect
Complete and balance the following chemical equations: i. CaCO3 + HCl - Sarthaks eConnect | Largest Online Education Community

How to Balance CaCO3 + HCl = CaCl2 + CO2 + H2O | How to Balance CaCO3 + HCl = CaCl2 + CO2 + H2O Hey there! Are you struggling with balancing

calcium carbonate reacts with HCl to give CaCl2 and CO2 according to reaction.what mass of 20% impure CaCO3 - Brainly.in

Study of CaCO3 particles formation kinetics using CaCl2 and Na2CO3: A.... | Download Scientific Diagram
Preparation of Ultra-fine Calcium Carbonate by a Solvent-free Reaction using Supersonic Airflow and Low Temperatures
16. In a chemical reaction, caco3+2hcl= cacl2 +co2+h2o. 25ml hcl and 0.75M Calculate the amount of caco3




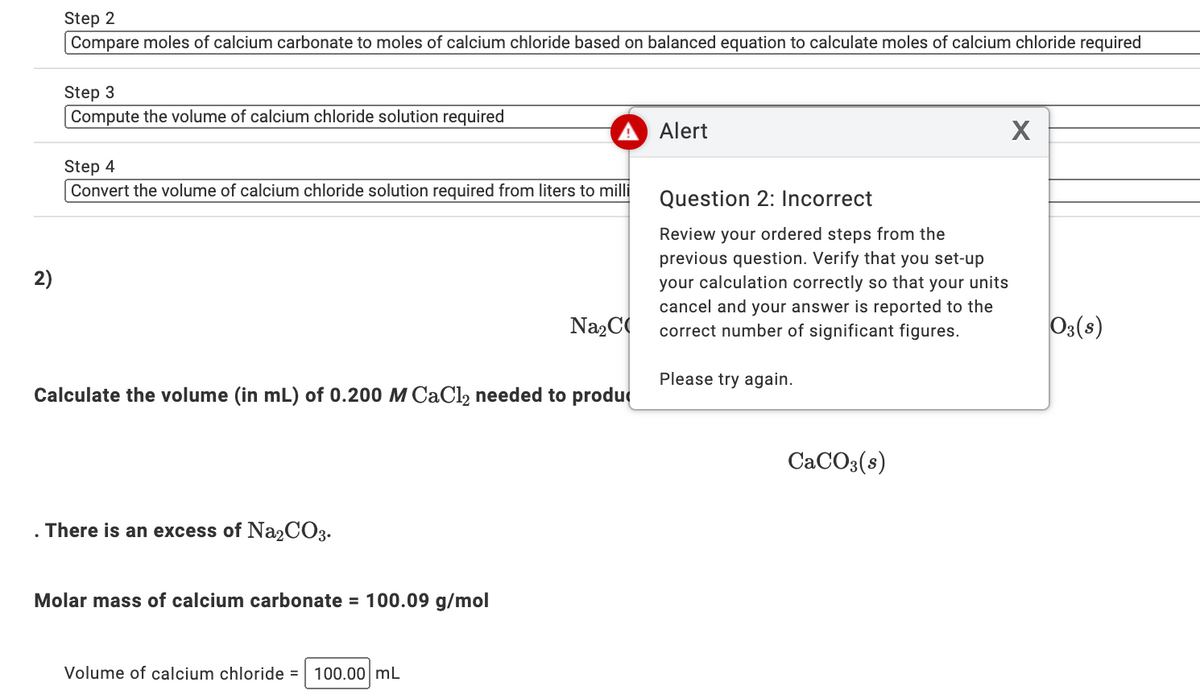



![ANSWERED] Na₂CO3(aq) +CaCl₂(aq) -> 2 NaCl(aq) + CaCO... - Organic Chemistry ANSWERED] Na₂CO3(aq) +CaCl₂(aq) -> 2 NaCl(aq) + CaCO... - Organic Chemistry](https://media.kunduz.com/media/sug-question/raw/61173905-1656941477.49281.jpeg)