Solubility of CO 2 in Water as a Function of the pH Value, At Indicated... | Download Scientific Diagram

Solubility of Carbon Monoxide and Hydrogen in Methanol and Methyl Formate: 298–373 K and 0.3–3.3 MPa | Journal of Chemical & Engineering Data

Solubility of Carbon Monoxide and Hydrogen in Methanol and Methyl Formate: 298–373 K and 0.3–3.3 MPa | Journal of Chemical & Engineering Data
![PDF] Solubility of hidrogen and carbon monoxide in water and some organic solvents | Semantic Scholar PDF] Solubility of hidrogen and carbon monoxide in water and some organic solvents | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/53e0b65259c80d553fe7dbb1371573c0d2fa02be/2-Figure1-1.png)
PDF] Solubility of hidrogen and carbon monoxide in water and some organic solvents | Semantic Scholar

Solubility of Carbon Monoxide and Hydrogen in Methanol and Methyl Formate: 298–373 K and 0.3–3.3 MPa | Journal of Chemical & Engineering Data
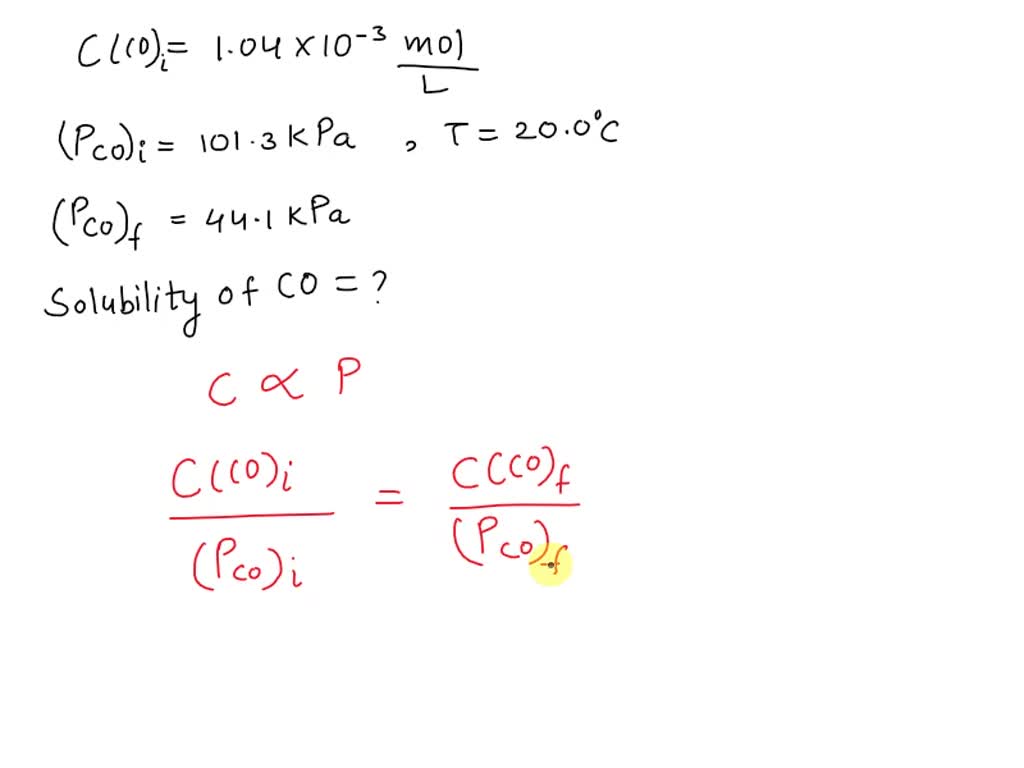
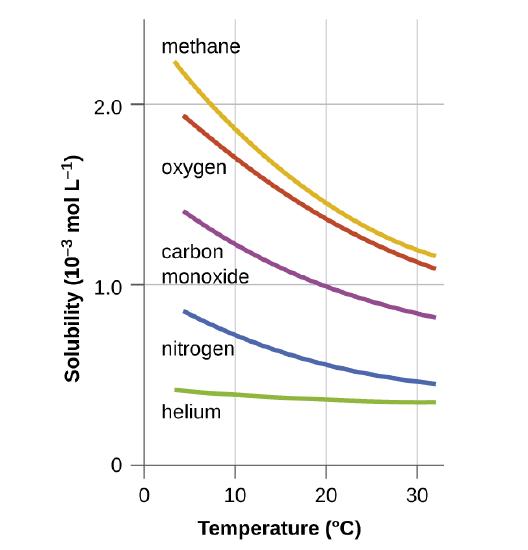
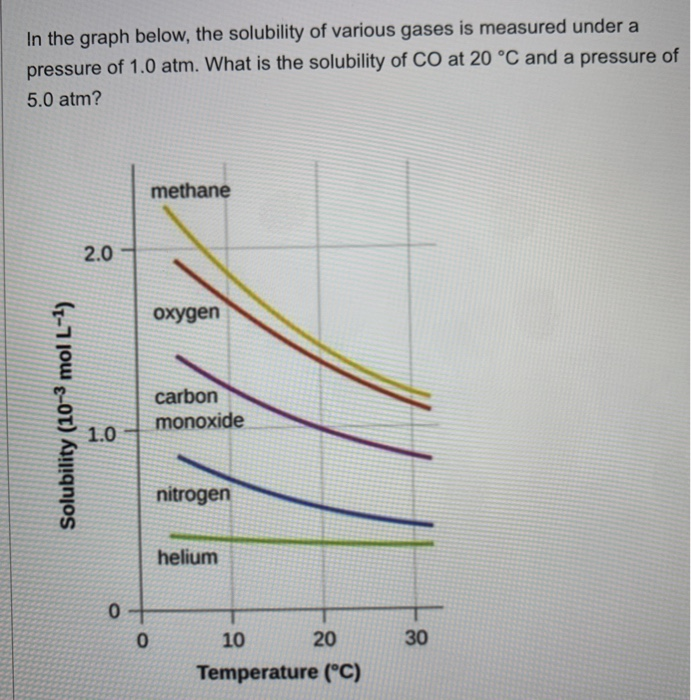
SOLVED: The concentration of dissolved carbon monoxide in water exposed to gaseous carbon monoxide at a partial pressure of 101.3 kPa at 20.0∘C is 1.04×10−3molL. Use Henry's law to determine the solubility
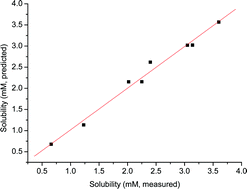
Carbon monoxide solubility in ionic liquids: determination, prediction and relevance to hydroformylation - Chemical Communications (RSC Publishing)

Buy Solubility Data Series: Carbon Monoxide (Volume 43) (IUPAC Solubility Data S., Volume 43) Book Online at Low Prices in India | Solubility Data Series: Carbon Monoxide (Volume 43) (IUPAC Solubility Data

SOLVED: Carbon monoxide gas is found to be more soluble in water than nitrogen gas, even though carbon monoxide and nitrogen have very similar Lewis structures. Why? What does having similar Lewis