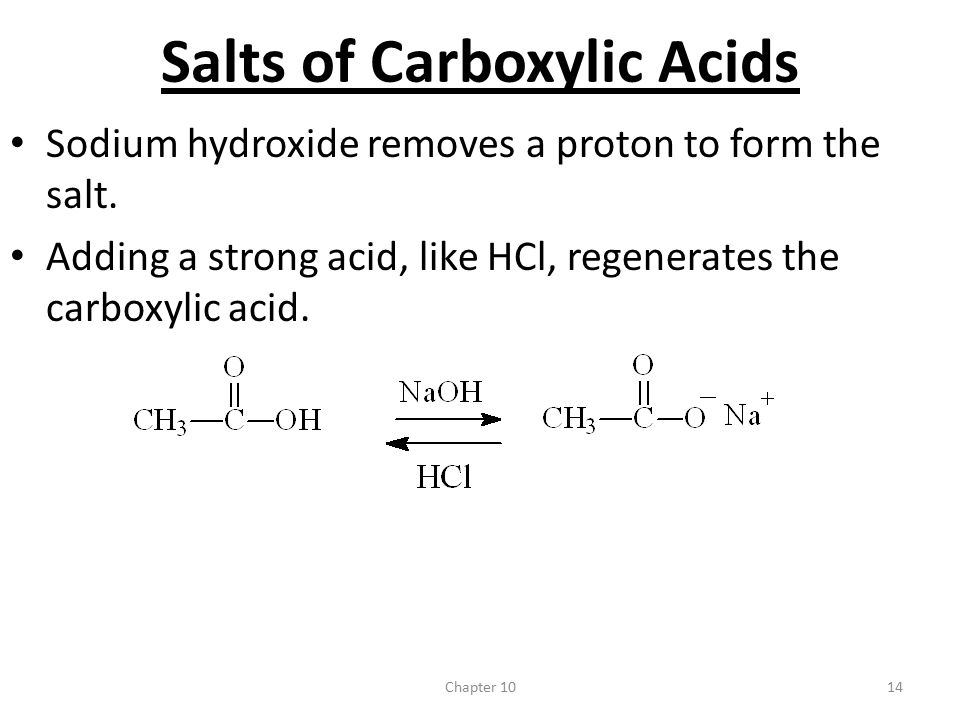
Chapter 10 Carboxylic Acids 1Chapter Introduction Carbonyl (-C=O) and hydroxyl (-OH) on the same carbon is carboxyl group. Carboxyl group is usually. - ppt download

Write a chemical equation for the conversion of the following carboxylic acid salt to its parent carboxylic acid. Let hydrochloric acid (HCl) be the source of the needed hydronium ions. Sodium lactate.

Relative Acidities of Carboxylic Acids, Phenols & Alcohols (7.5.2) | CIE A Level Chemistry Revision Notes 2019 | Save My Exams

Write a chemical equation for the conversion of the following carboxylic acid salt to its parent carboxylic acid. Let hydrochloric acid (HCl) be the source of the needed hydronium ions. Sodium lactate.
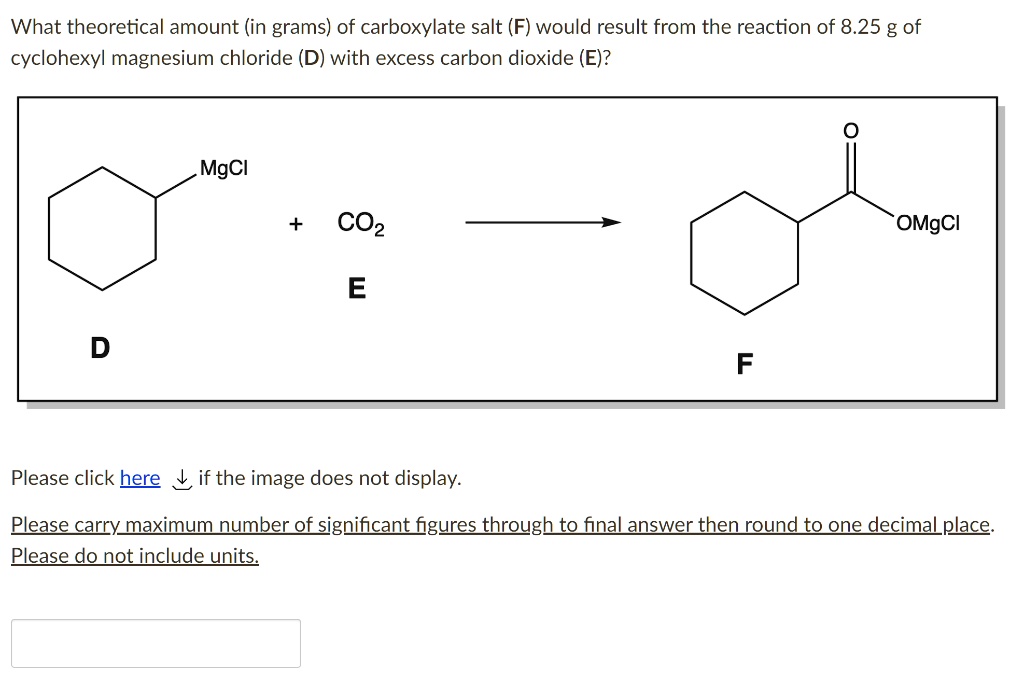
SOLVED: What theoretical amount (in grams) of carboxylate salt (F) would result from the reaction of 8.25 g of cyclohexyl magnesium chloride (D) with excess carbon dioxide (E)? MgCl OMgCl Please click
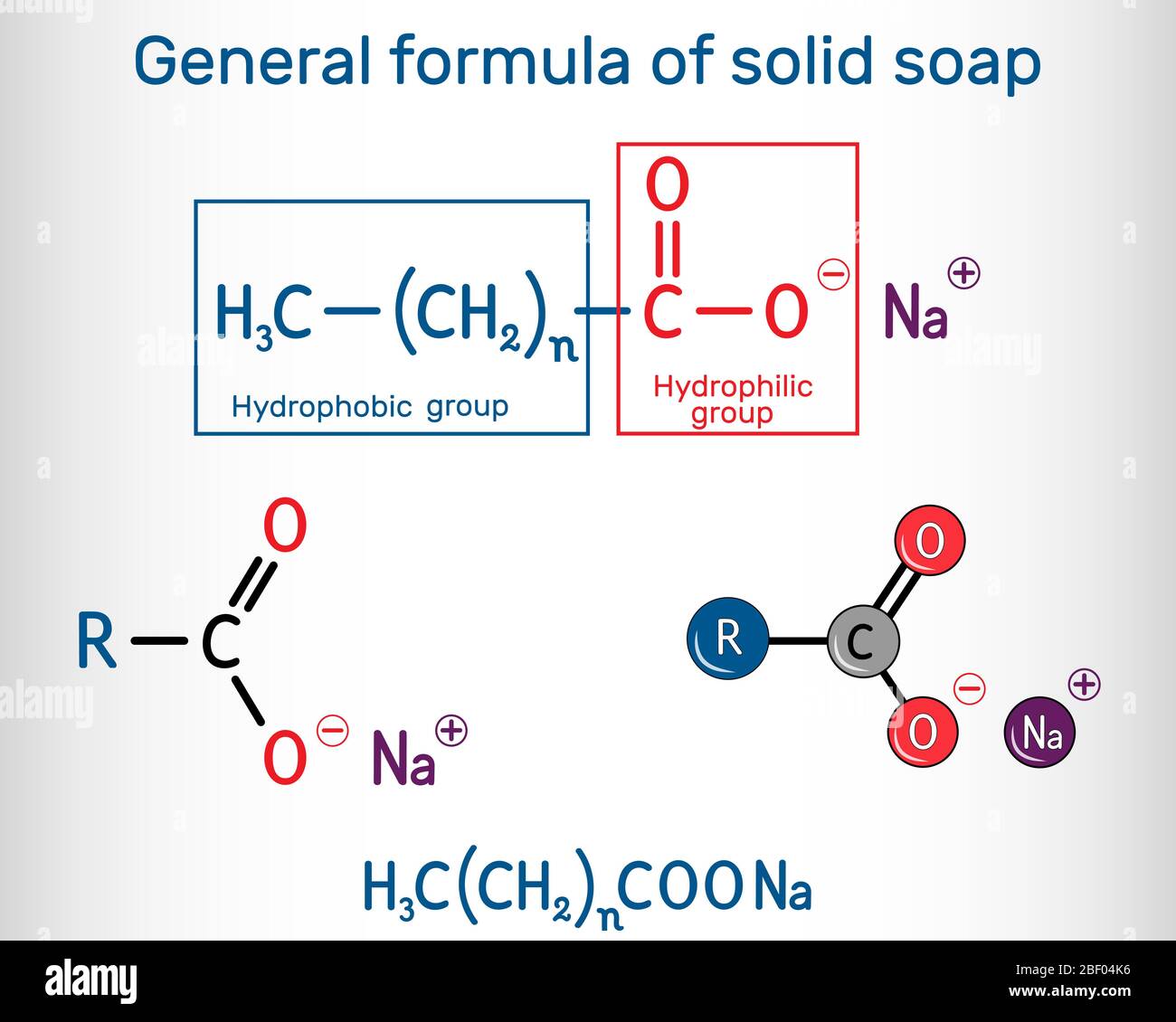
General formula of solid soap molecule. Sodium carboxylate, RCOONa. It is the sodium salt of fatty acid. Structural chemical formula and molecule mode Stock Vector Image & Art - Alamy





![Deprotonation [carboxylate formation] - ChemistryScore Deprotonation [carboxylate formation] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Deprotonation-carboxylate-formation2-768x252.png)

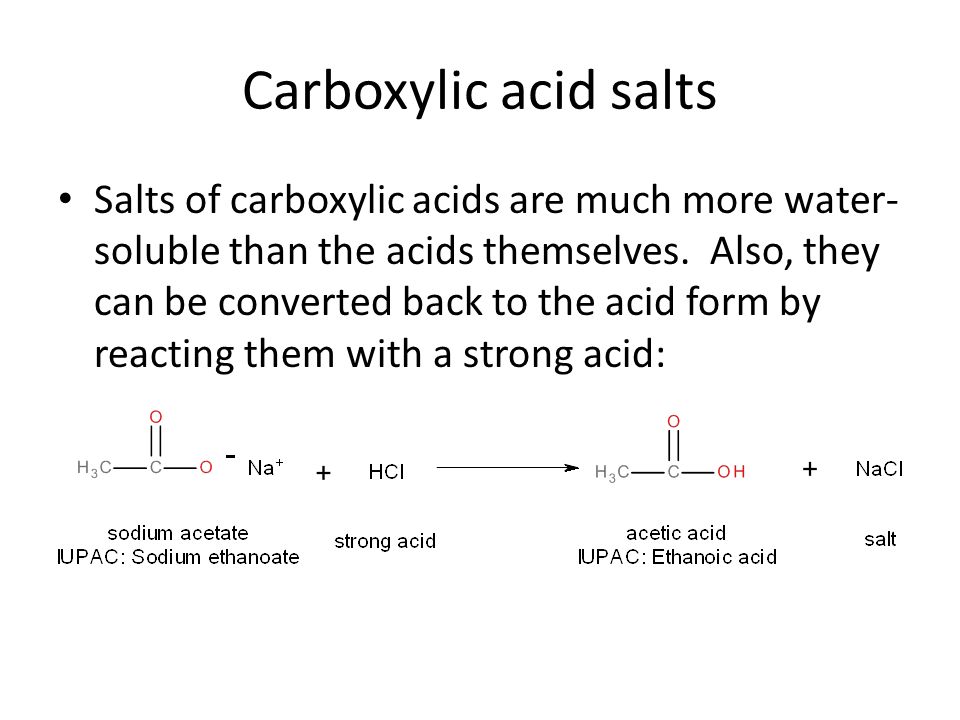




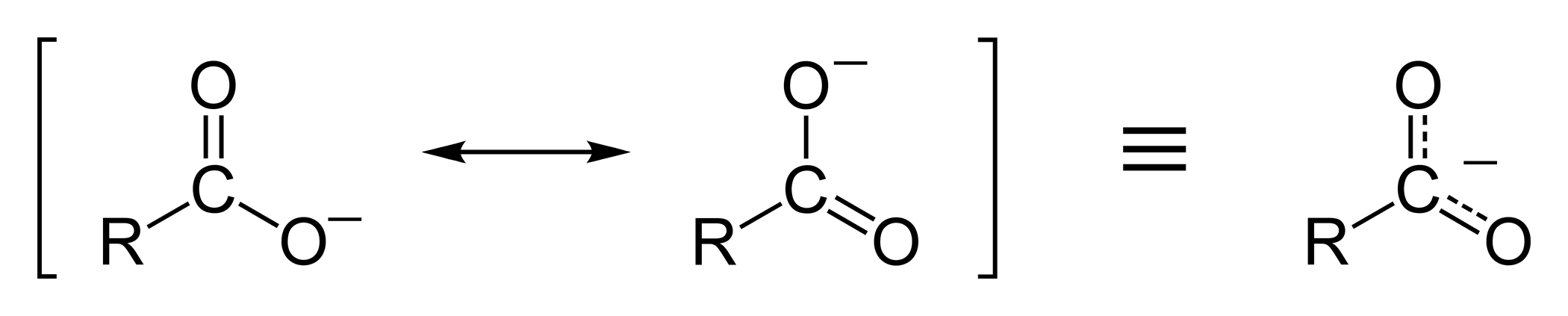
![Deprotonation [carboxylate formation] - ChemistryScore Deprotonation [carboxylate formation] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Deprotonation-carboxylate-formation1-768x276.png)


