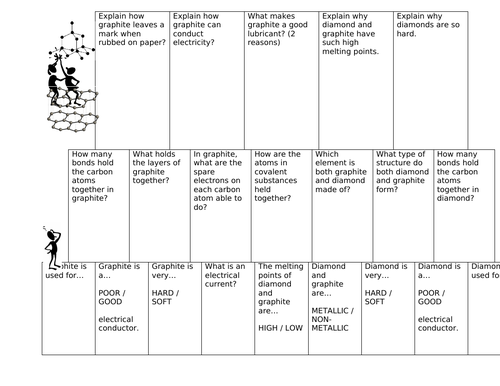
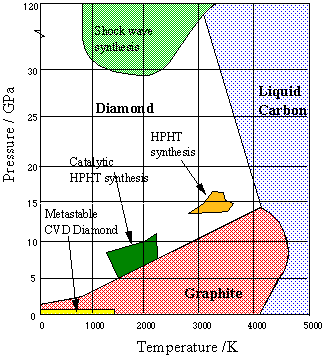
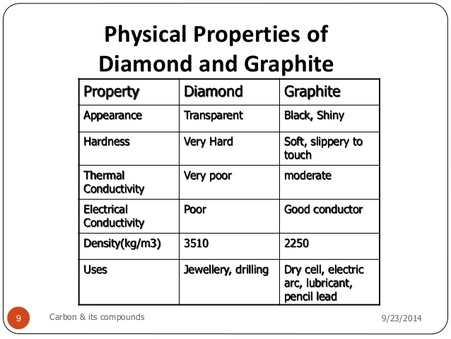
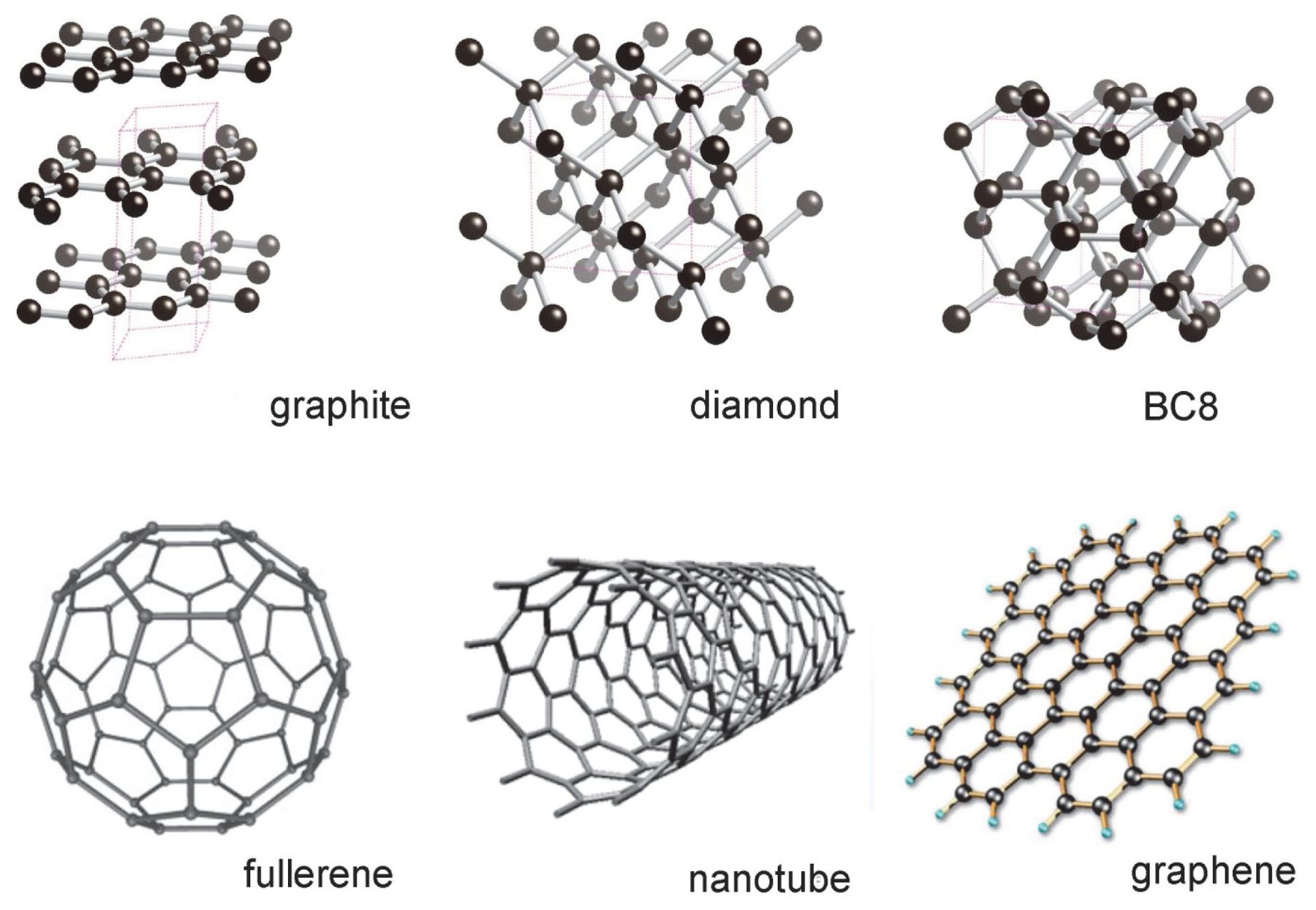
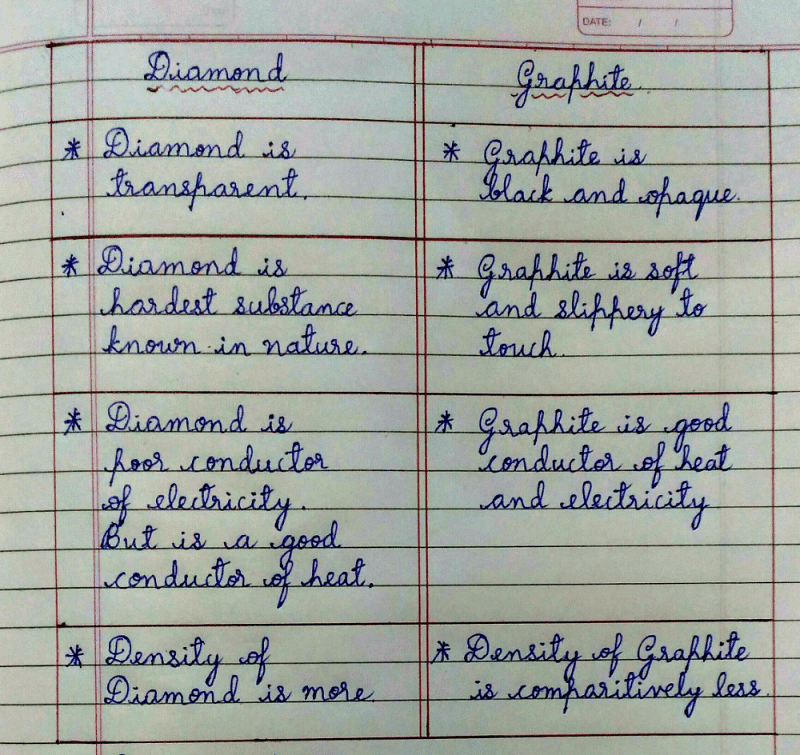
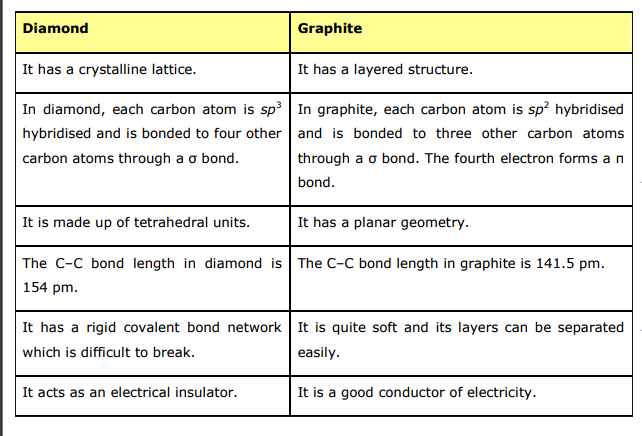
Explain the difference: A. Diamond and graphite. B. Crystalline and non-crystalline forms of carbon.
Q.Asseration:Diamond and graphite do not have the same crystal structure. Reason: Diamond is crystalline while graphite is amorphous
Explain the difference in properties of diamond and graphite on the basis of their structures. - Sarthaks eConnect | Largest Online Education Community

UNM Foundation Engineering - Do you know that diamond and graphite are made from the same element? Source: https://www.bbc.co.uk/bitesize/guides/zspxdxs/revision/2 . . . . . . #WeAreUoN #UoNMalaysia #UNM #nottinghammalaysia #nottinghamuniversity ...