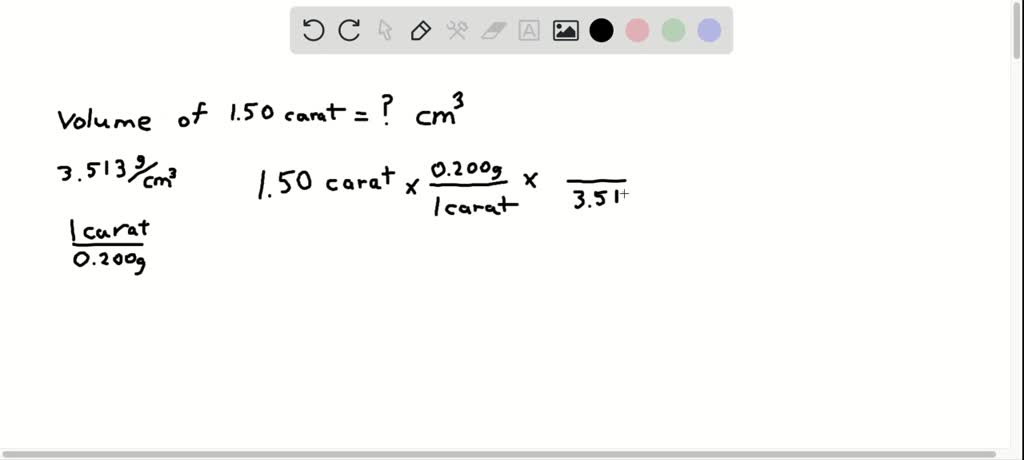
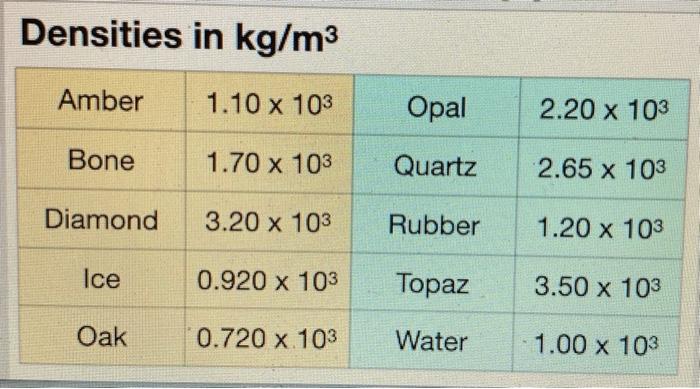
SOLVED:Diamond has a density of 3.513 g / cm^3 . The mass of diamonds is often measured in "carats," where 1 carat equals 0.200 g . What is the volume (in cubic

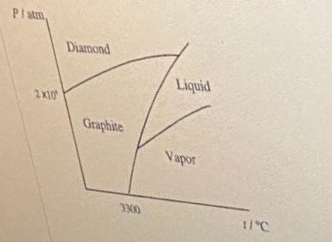
The densities of graphite and diamond at `298K` are `2.25` and `3.31gcm^(-3)` , respectively - YouTube
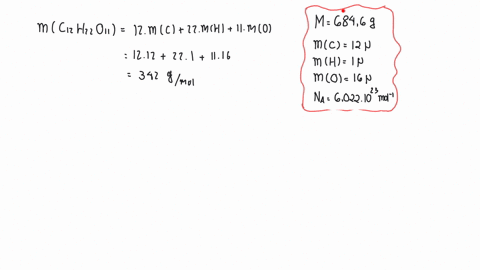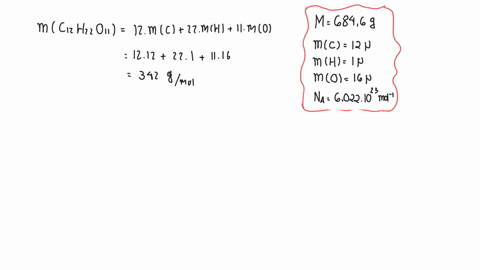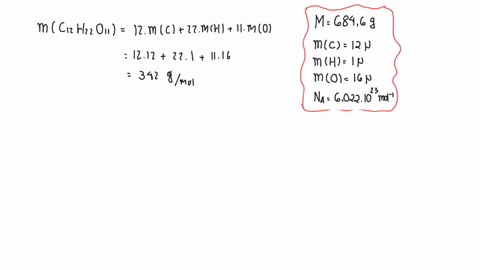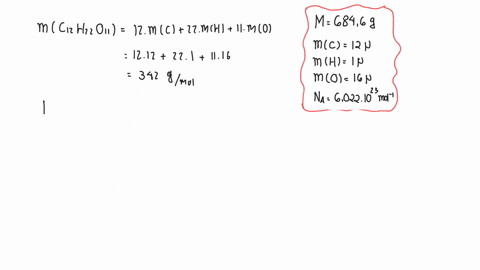
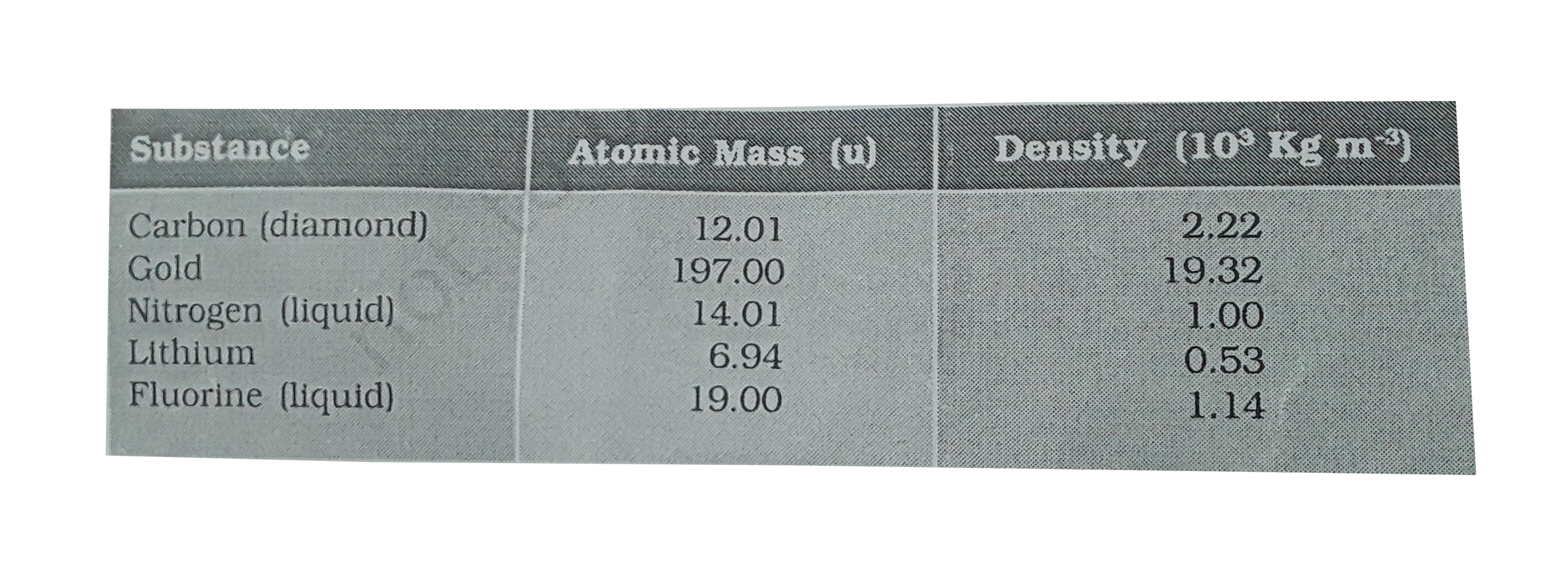
SOLVED:The mass density of diamond (a crystalline form of carbon) is 3500 kg / m^3 . How many carbon atoms per cm^3 are there?



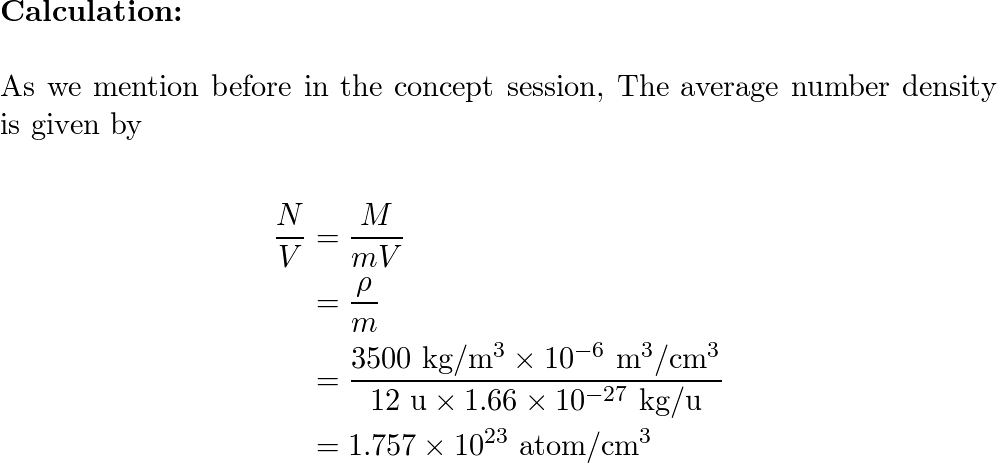





![Liquid steel viscosity [13] | Download Scientific Diagram Liquid steel viscosity [13] | Download Scientific Diagram](https://www.researchgate.net/profile/Zbigniew-Malinowski/publication/267448472/figure/fig3/AS:295601850863617@1447488381205/Liquid-steel-viscosity-13_Q320.jpg)




![Liquid steel density [14] | Download Scientific Diagram Liquid steel density [14] | Download Scientific Diagram](https://www.researchgate.net/publication/267448472/figure/fig4/AS:295601850863618@1447488381354/Liquid-steel-density-14.png)