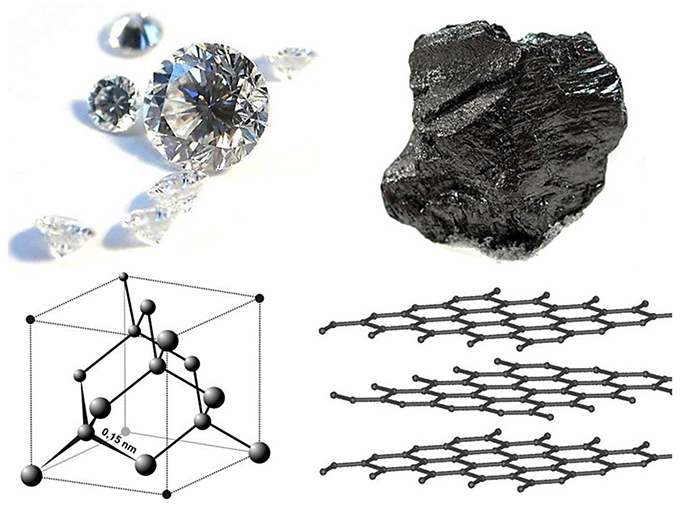
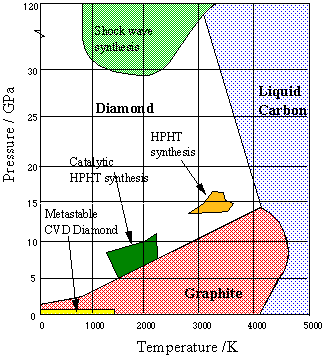
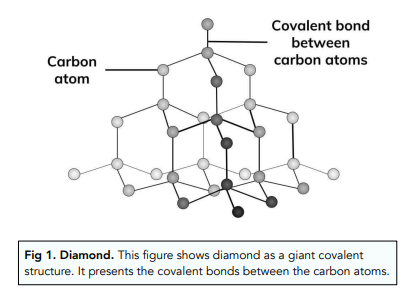
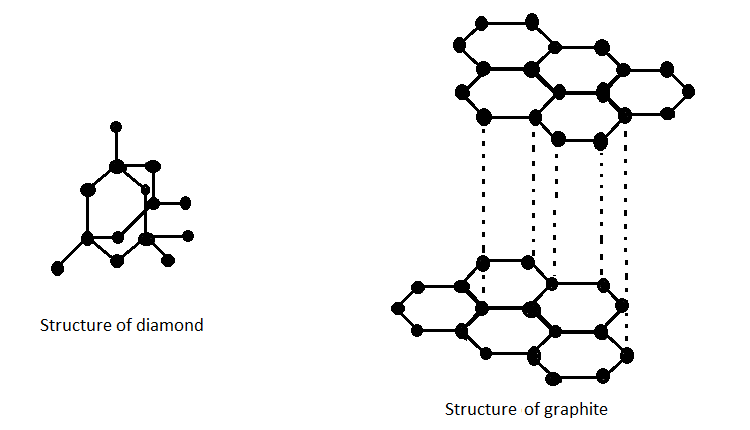
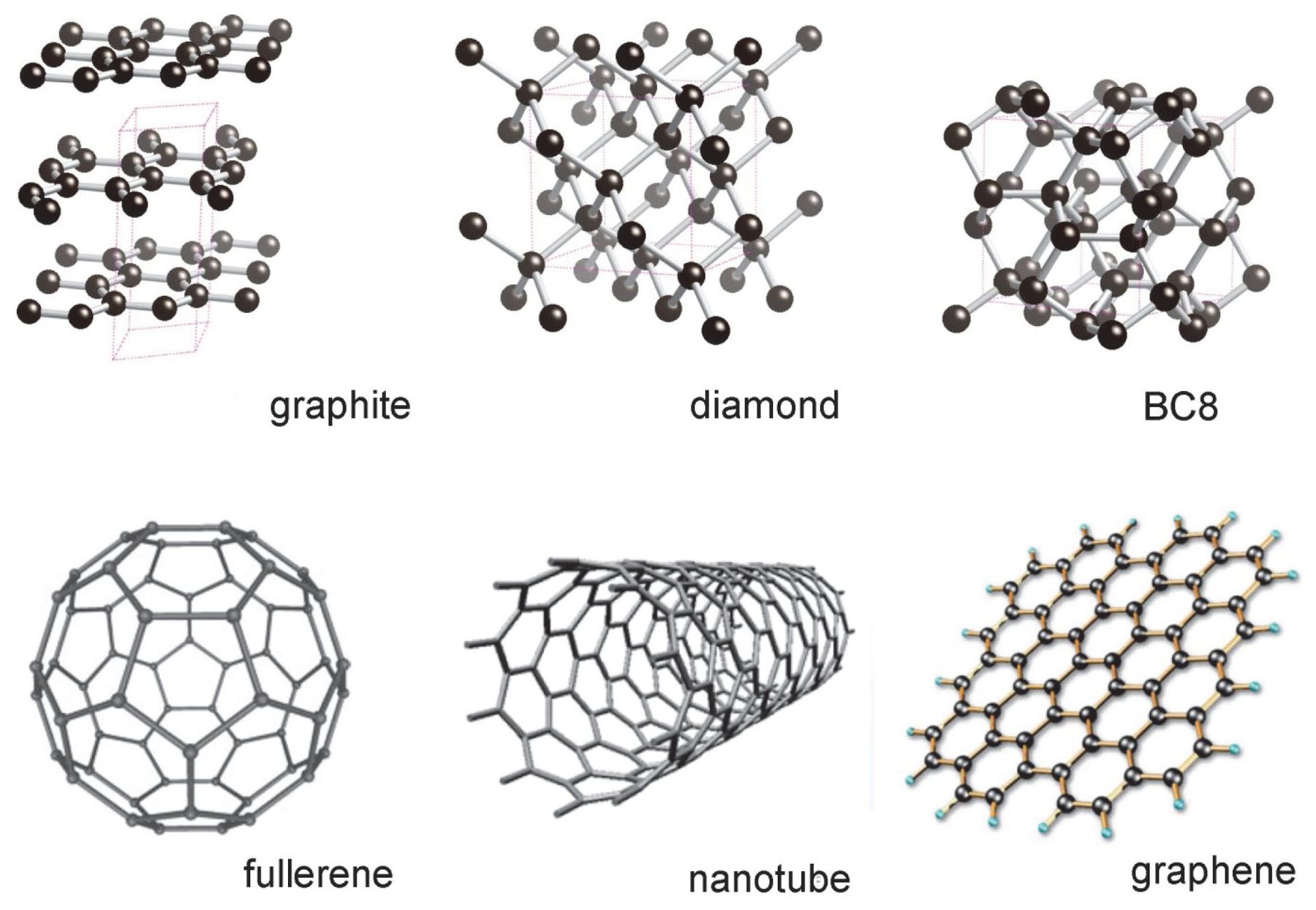
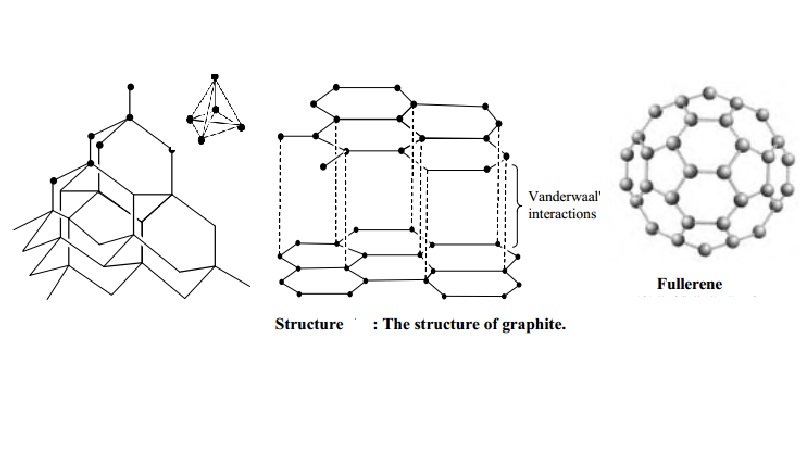
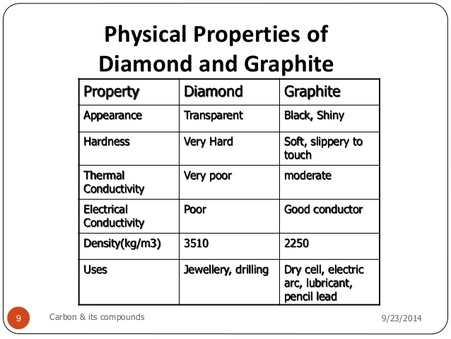
CARBON – From the element of life to advanced technology _ PART I (WHY is Carbon special?) – Computer Aided Design & The 118 Elements
![Transformation of diamond structure to more stable graphite structure [40]. | Download Scientific Diagram Transformation of diamond structure to more stable graphite structure [40]. | Download Scientific Diagram](https://www.researchgate.net/publication/337644533/figure/fig4/AS:830761544462336@1575080395347/Transformation-of-diamond-structure-to-more-stable-graphite-structure-40.jpg)
Transformation of diamond structure to more stable graphite structure [40]. | Download Scientific Diagram
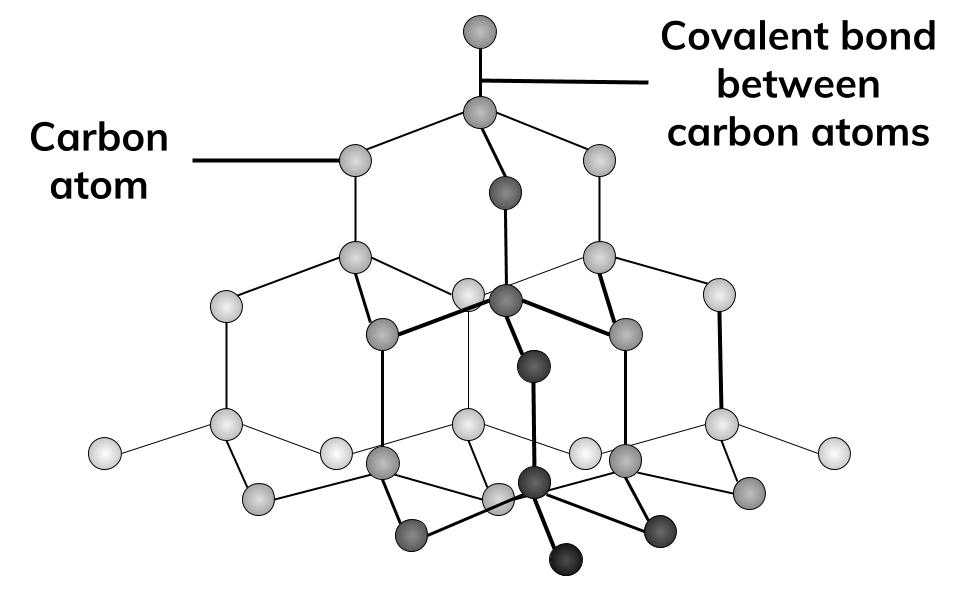
1:50 explain how the structures of diamond, graphite and C60 fullerene influence their physical properties, including electrical conductivity and hardness - TutorMyself Chemistry
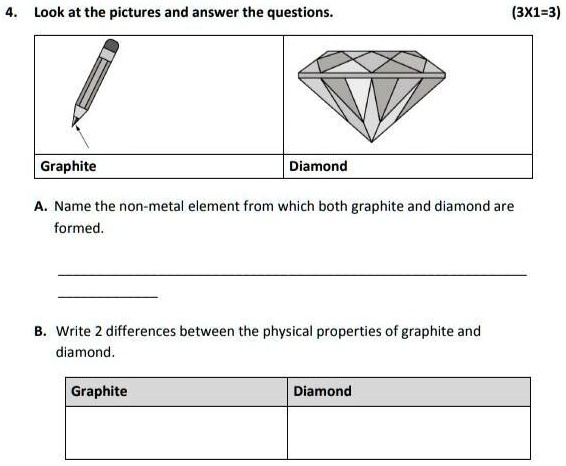
SOLVED: "please answer don't spam Look at the pictures and answer the questions (3X1-3) Graphite Diamond Name the non-metal element from which both graphite and diamond are formed Write 2 differences between

Sevenoaks SciTech on Twitter: "Element of the Week: #Carbon - atomic number 6. Occurs in three forms, #diamond, #graphite and #fullerenes. http://t.co/RImH1lmmDH" / Twitter