The combustion enthalpies of carbon, hydrogen, and methane are 395.5 kJ mol^ 1, 284.8 kJ mol^ 1 and 890.4 kJ mol^ 1 respectively at 25^0C. The value of s†an dard formation enthalpies

The enthalpy of combustion of carbon, hydrogen and sucrose are –393.5, –286.2 and –5644.2 kJ/mol - Brainly.in
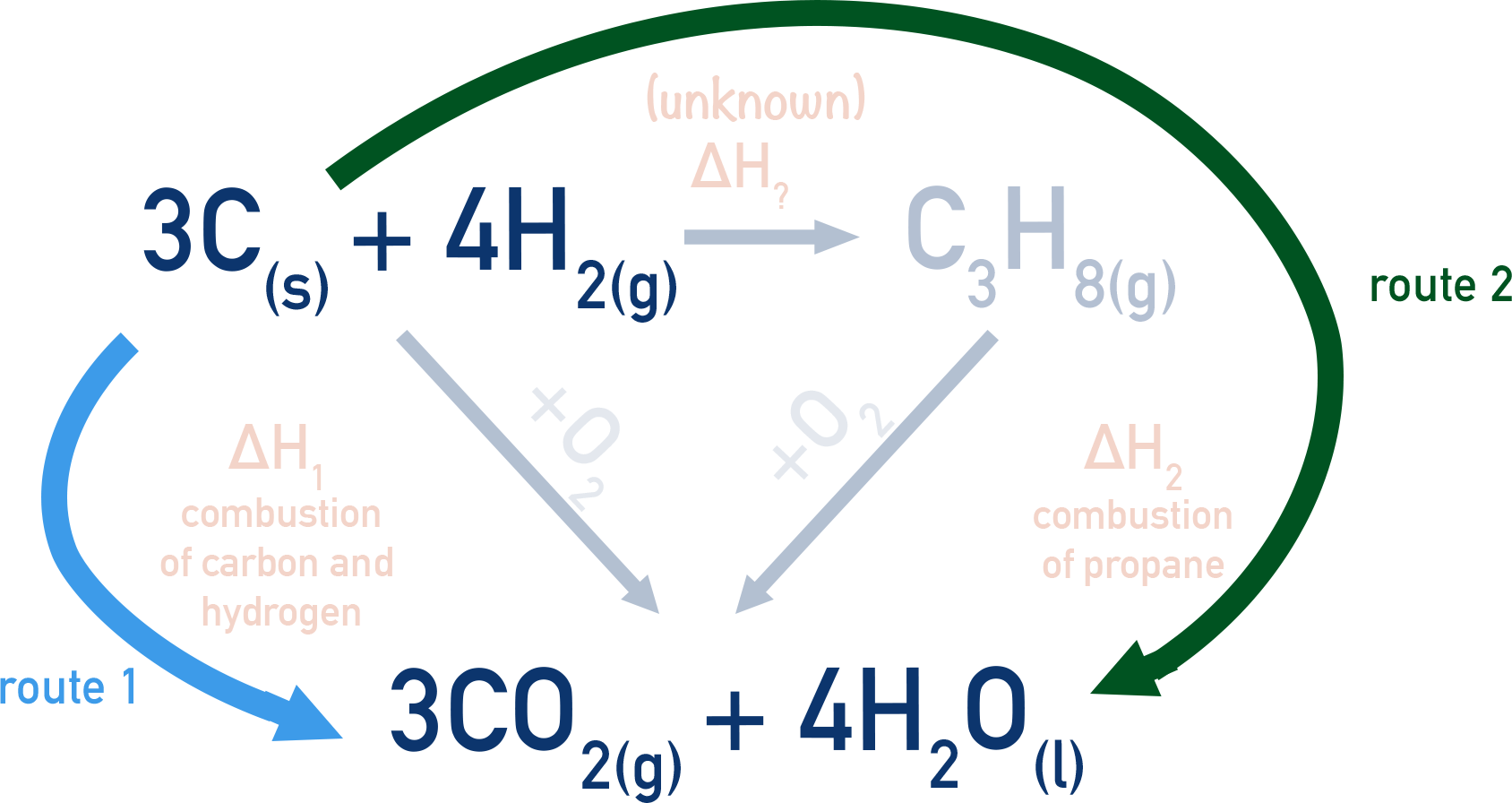
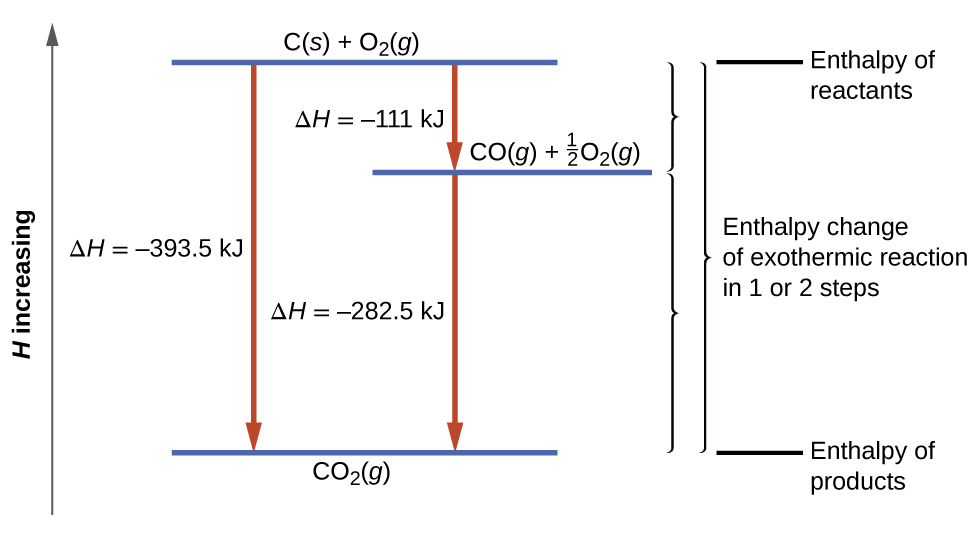
The enthalpies of combustion of carbon and carbon monoxide are -393.5 and –283kJ mol^–1 respectively. - Sarthaks eConnect | Largest Online Education Community

The enthalpies of combustion of carbon and carbon monoxide are `-390 kJ mol^(-1)` and `-278 kJ mo - YouTube

Calculate the standard heat of formation of carbon disulphide (l). Given that the standard heats of - YouTube

41524992Given standard enthalpy of formation of `CO(-110 \"KJ mol\"^(-1))` and `CO_(2)(-394 \"KJ mol - YouTube

The combustion enthalpies of carbon, hydrogen and methane are 395.5 kJ/mol , - 285.8 kJ/mol and - 890.4kJ/mol respectively at 25^∘C . The value of standard formation enthalpies of methane at that temperature is:

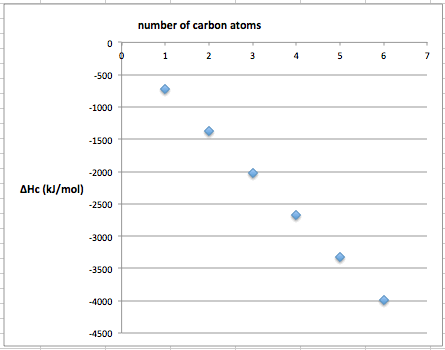
A level Enthalpy data patterns combustion alkanes & alcohols, bond enthalpy, bond length, bond order, enthalpy of combustion for a homologous series KS5 GCE A level chemistry revision notes

Enthalpy of combustion of carbon to CO2 is -393.5KJ mol-1. Calculate the heat released upon..... - YouTube

Enthalpy of combustion of carbon to CO2 is - 393.5 KJ/mole. The heat released upon the formation of 35.2g of CO2 from carbon and dioxygen gas is.

The standard heat of formation of carbon disulphide (l) given that standard heat of combustion of - YouTube
What is the enthalpy of formation of carbon monoxide in KJ/mol ? C(s) + O2(g) --> CO2(g) ΔH° = -393 kJ 2CO(g) + O2(g) --> 2CO(g) ΔH° = -588 kJ | Socratic
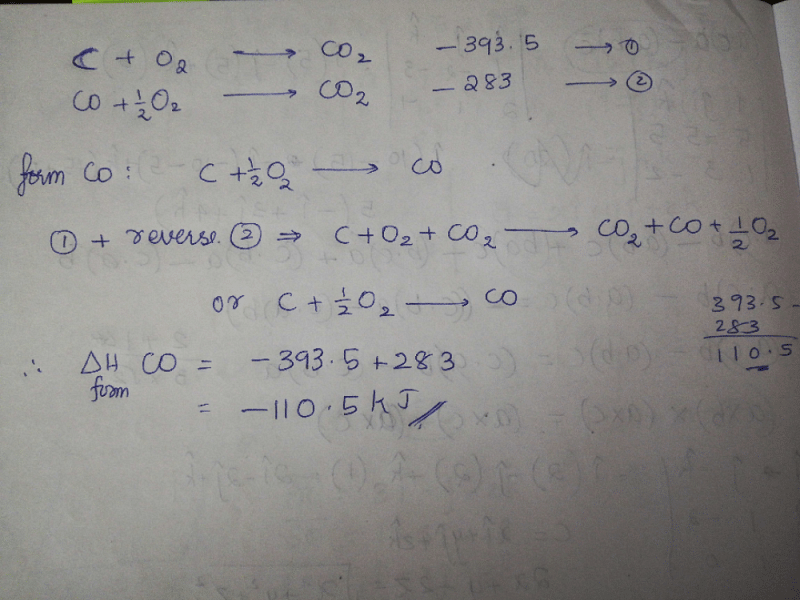
The enthalpies of combustion of carbon and carbon monoxide are -393.5 and -283 KJ mol¹ respectively .The enthalpy of formation of carbon monoxide per mol isa)-110.5 KJb)-676.5 KJc)+676.5 KJd)110.5 KJCorrect answer is

Question Video: Calculating Standard Enthalpy of Combustion of Methane Using Standard Enthalpies of Formation of Methane and Carbon Dioxide | Nagwa
62.The Enthalpies of combustion of carbon and carbon monoxide are 390 kJ and 278kJ respectively. The enthalpy of formation of carbon monooxide is? a) 669 kJ b) 112 kJ c) 112 kJ d) 668 kJ
![Enthalpy of combustion of carbon to CO2 is - 393.5 kJmol^-1 . The heat released upon formation of 35.2 g of CO2 from carbon and dioxygen gas is: [Molar mass of CO2 = 44 gmol^-1 ] Enthalpy of combustion of carbon to CO2 is - 393.5 kJmol^-1 . The heat released upon formation of 35.2 g of CO2 from carbon and dioxygen gas is: [Molar mass of CO2 = 44 gmol^-1 ]](https://haygot.s3.amazonaws.com/questions/1961395_1291143_ans_d6642064cda84dfe904274b61e90a2dc.jpg)
Enthalpy of combustion of carbon to CO2 is - 393.5 kJmol^-1 . The heat released upon formation of 35.2 g of CO2 from carbon and dioxygen gas is: [Molar mass of CO2 = 44 gmol^-1 ]