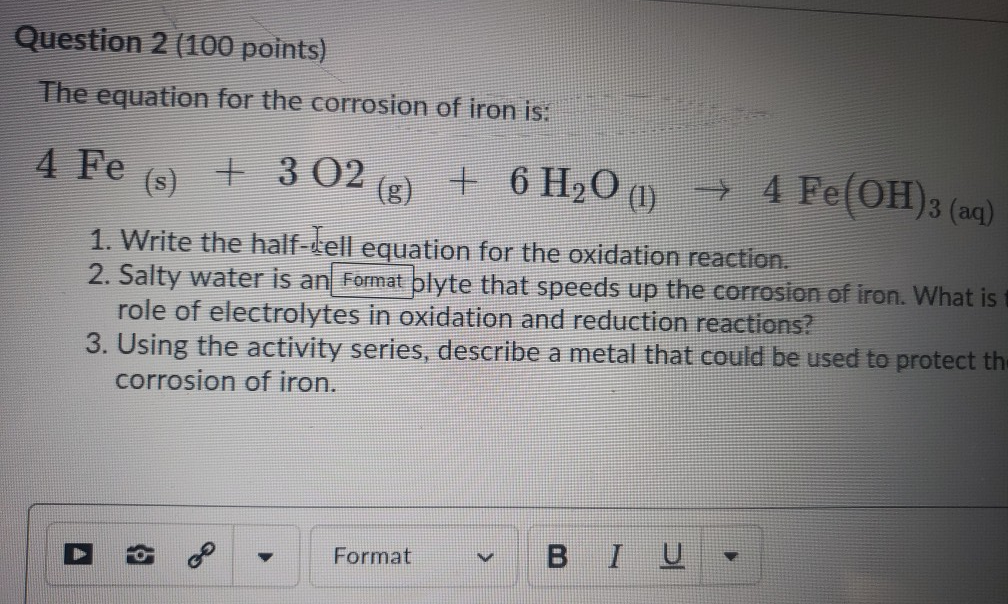
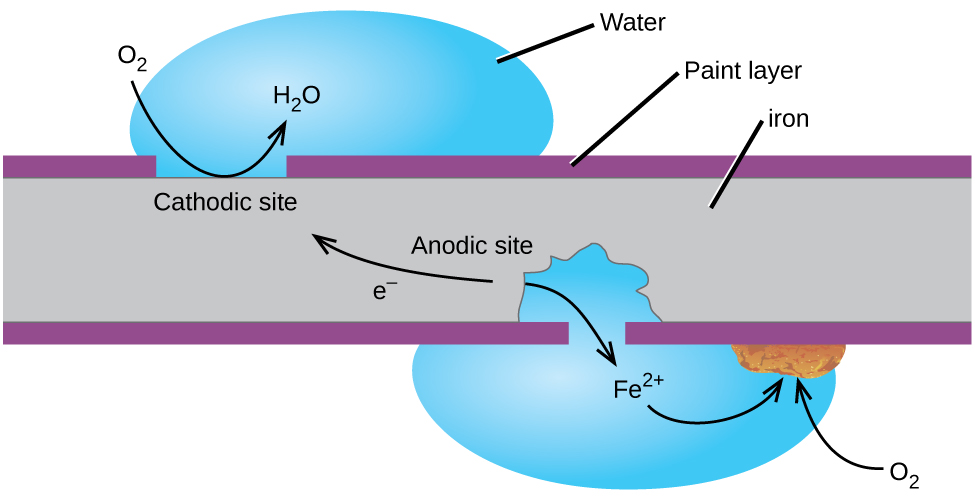
Iron + Water = Rust | Chemistry! | Balancing Equations, Coefficients, Molecules - Science Q&A - YouTube

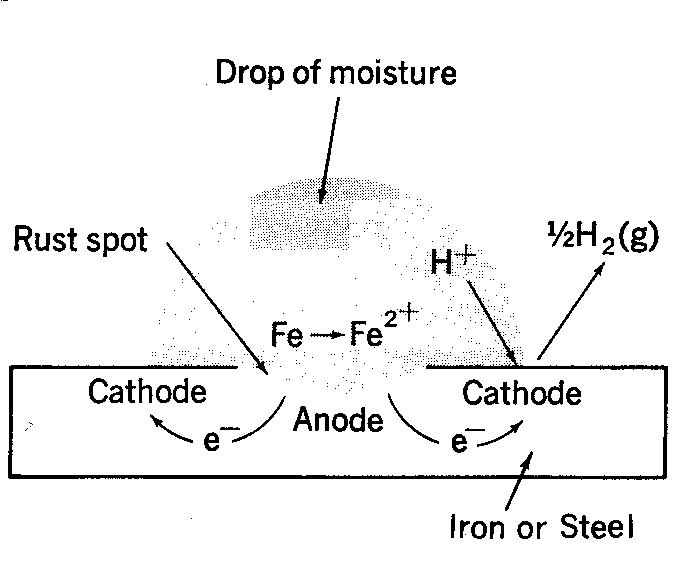
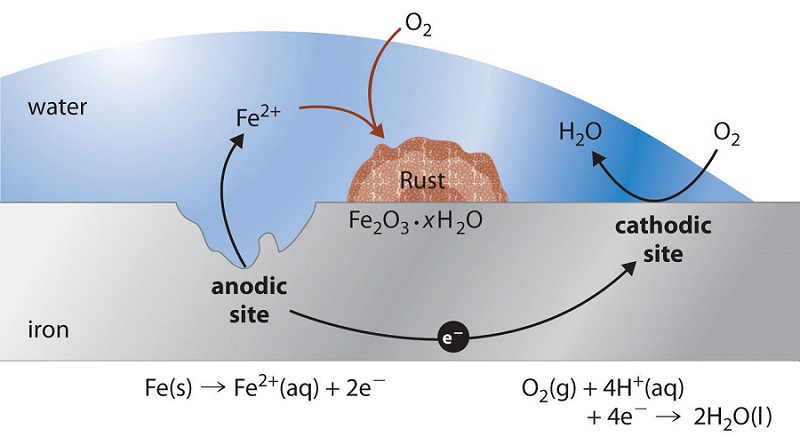
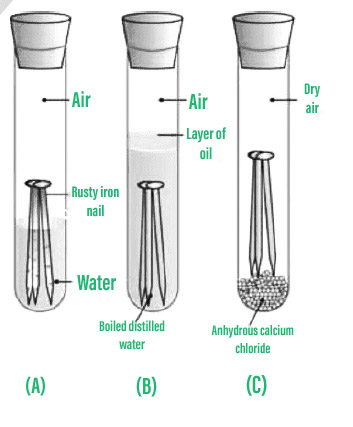
Causes chemistry of rusting rust prevention introduction to oxidation reduction REDOX reactions gcse igcse KS4 science chemistry revision notes revising

Translate the following statements into chemical equations and then balance them:(a) Hydrogen gas combines with nitrogen to form ammonia.(b) Hydrogen sulphide gas burns in air to give water and sulphur dioxide.(c) Barium
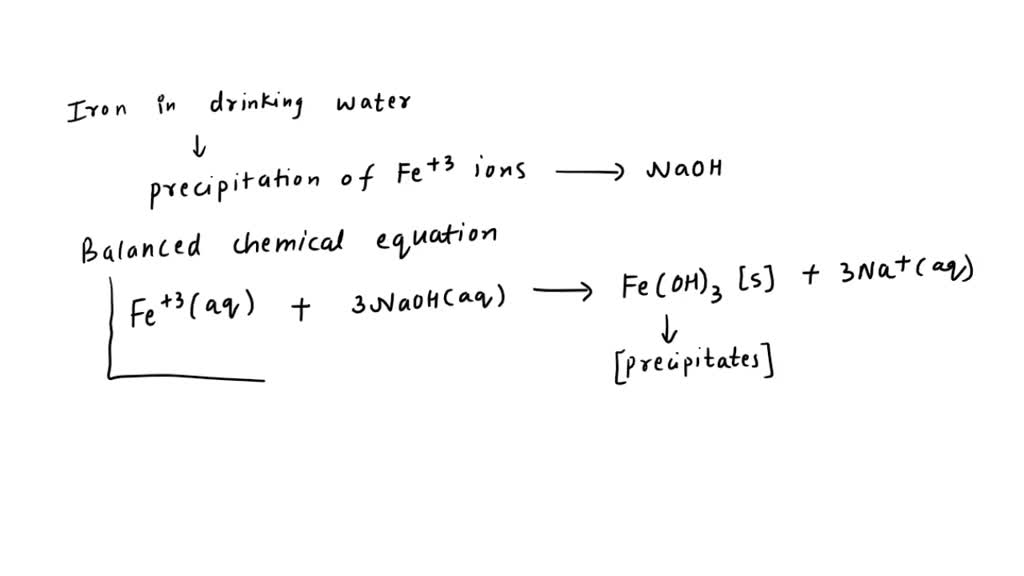
SOLVED: Iron in drinking water is removed by precipitation of the Fe3+ ion by reaction with NaOH to produce iron(III) hydroxide. Write the balanced chemical equation and the net ionic equation for
Give balanced chemical equations for the reaction of water with (a) Sodium (b) Iron - Sarthaks eConnect | Largest Online Education Community