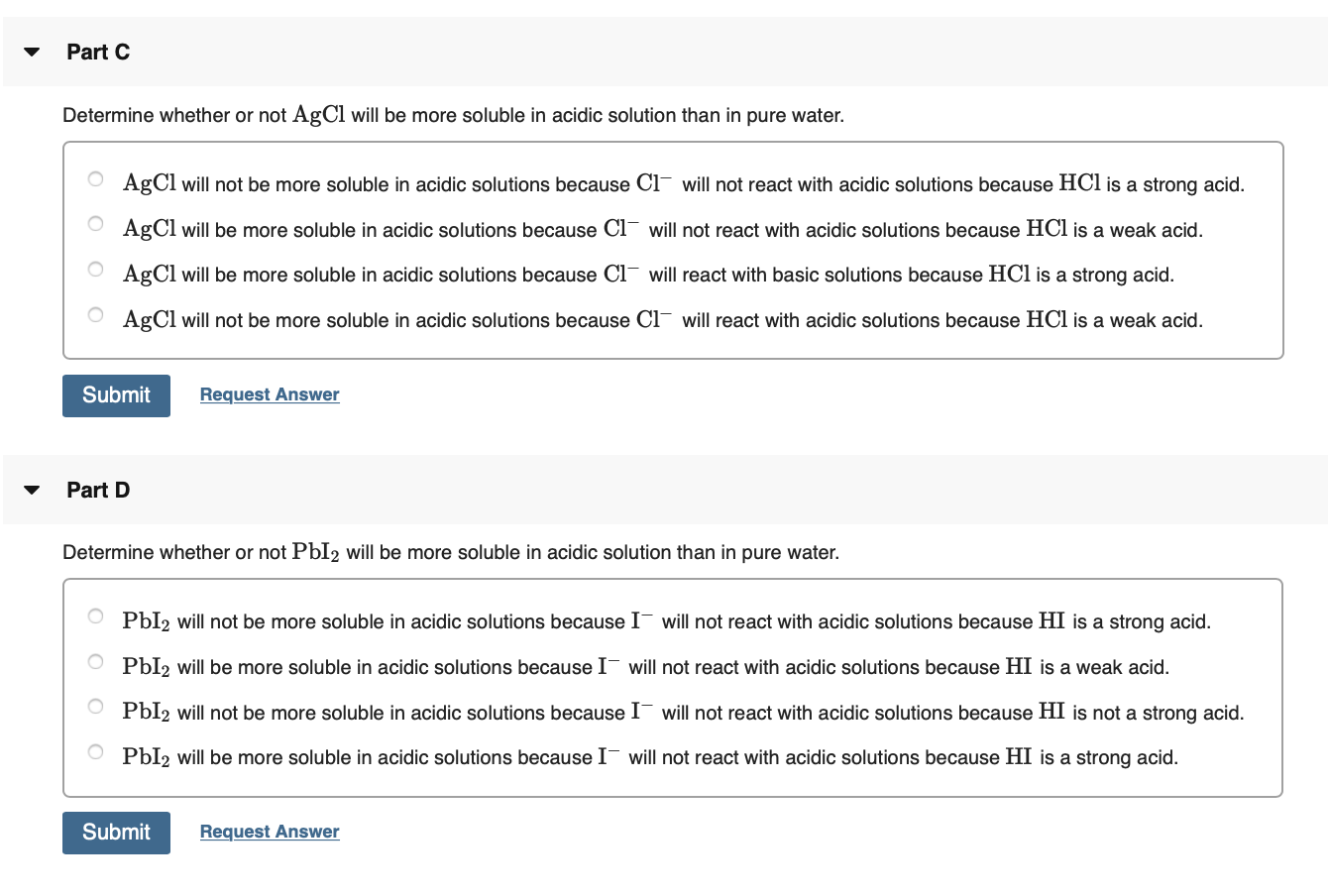
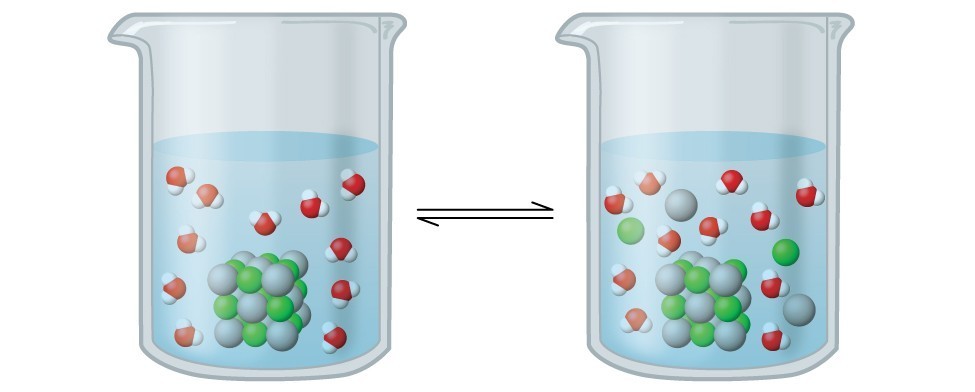
Solubility Product Constants Silver chloride, AgCl,is rather insoluble in water. Careful experiments show that if solid … | Solubility, Silver chloride, Experiments

Solubility Product Constants Silver chloride, AgCl,is rather insoluble in water. Careful experiments show that if solid AgCl is placed in pure water and. - ppt download

Question Video: Using Precipitation Gravimetry to Calculate the Percentage Mass of NaCl in a Mixed Sample | Nagwa

Procedure method making insoluble salt by precipitation reaction from two soluble compounds apparatus chemicals procedures equations use of barium sulfate meal gcse chemistry KS3 KS4 Science IGCSE O level revision notes

AgCl) Silver Chloride with High Purity Silver Plating CAS 7783-90-6 - China Silver Chloride, 7783-90-6 | Made-in-China.com

Unit cell representation for the rock salt structures of (A) AgCl and... | Download Scientific Diagram

The Solubility Product Principle. 2 Silver chloride, AgCl,is rather insoluble in water. Careful experiments show that if solid AgCl is placed in pure. - ppt download
2.7 Construction of a silver/silver chloride reference electrode with a... | Download Scientific Diagram

SOLVED: Silver chloride crystallizes with the sodium chloride (rock salt) structure. the length of a unit cell edge is 555 pm. what is the density of agcl?

SOLVED: QUESTION 1 What is the solubility of AgCl (in M): (a) in pure water and (b) in a 3.4 x 10-3 M AgNO3 Note: AgCl (s) <–> Agt (aq) + Cl (